 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
| Cat. No. | Product Name | Field of Application | Chemical Structure |
|---|---|---|---|
| DC67990 | 4A3-LNSC8 Featured |
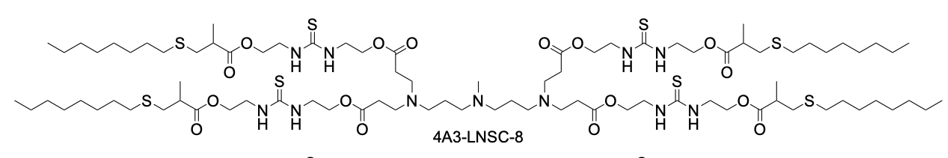
4A3-LNSC8 is a strategically designed thiourea-functionalized ionizable lipid that serves as the foundational core for a novel anion-coordination delivery platform. Its structure features a central 4A3 amine headgroup symmetrically extended with four hydrophobic tails, each incorporating a biodegradable ester linkage and a key thiourea-bridged linker. The inclusion of the thiourea group is the pivotal innovation, as it provides specific hydrogen-bonding sites capable of interacting with various halide anions (F⁻, Cl⁻, I⁻). When formulated into lipid nanoparticles (LNPs) without anion coordination, 4A3-LNSC8 itself exhibits a characteristic liver tropism, efficiently delivering mRNA to hepatocytes following systemic administration, with a measured surface pKa of approximately 5.54. However, its primary significance lies in its role as a versatile precursor. The strong anion-binding capability of its thiourea linkers allows for predictable modulation of the LNP's properties. Upon binding with anions like Cl⁻, the resulting complex (e.g., Cl-4A3-LNSC8) undergoes a significant pKa shift, which reprograms the LNP's in vivo fate, redirecting mRNA delivery from the liver to secondary lymphoid organs such as the spleen and lymph nodes. Thus, 4A3-LNSC8 is not merely an efficient ionizable lipid but a programmable scaffold that enables precise control over organ-targeting specificity through simple anion coordination, offering a powerful rational design strategy for advanced mRNA therapeutics.
More description
|
|
| DC67989 | Cl-4A3-LNSC8 Featured |
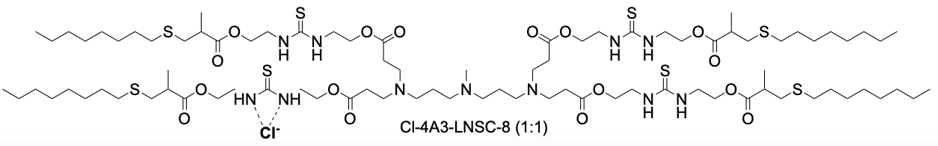
Cl-4A3-LNSC8 represents a novel class of thiourea-functionalized ionizable lipids engineered for selective organ-targeted mRNA delivery. Its core innovation lies in an anion-coordination strategy, where the parent lipid, 4A3-LNSC8, binds chloride ions (Cl⁻) via hydrogen-bonding interactions with its thiourea groups. This binding event is not merely structural but functionally critical, as it induces a significant shift in the surface pKa of the resulting lipid nanoparticles (LNPs) from approximately 5.54 to 8.79. This pKa modulation is the key mechanism that redirects the organotropism of the LNPs upon systemic administration. While the unmodified 4A3-LNSC8 LNPs preferentially deliver mRNA to the liver, Cl-4A3-LNSC8 LNPs effectivelyreprogram this tropism, enabling highly efficient mRNA delivery to secondary lymphoid organs (SLOs), particularly the spleen and lymph nodes. This platform demonstrates remarkable efficacy, achieving up to 65.7% gene editing efficiency in splenic macrophages in vivo, significantly outperforming benchmark delivery systems. Furthermore, by leveraging the coordination with different halides, such as iodine for computed tomography (CT) contrast, the system can be adapted for dual-modal theranostic applications, enabling simultaneous lymphatic metastasis imaging and therapeutic mRNA delivery.
More description
|
|
| DC67602 | ILB-3132(E12LA6B603) Featured |
E12LA6B603(ILB3132,ILB-3132) is a novel ionizable amino lipid disclosed in patent WO2024198497A1, developed by MagicRNA, representing a highly efficient component for lipid nanoparticle (LNP) delivery systems.When formulated into LNPs, E12LA6B603 LNP achieves a remarkable 98.26% encapsulation efficiency for mRNA. It mediates superior in vitro transfection in dendritic cells (1.8E+05 intensity) and demonstrates best-in-class in vivo protein expression after intramuscular injection (2.2E+09 intensity). Most notably, in a B16-OVA melanoma model, therapeutic OVA-mRNA vaccines delivered by E12LA6B603 LNPs induced 100% complete tumor regression, highlighting its superior efficacy over benchmarks like DLin-MC3 and SM-102. Its biodegradable ester linkages and balanced structure make it a promising, potent candidate for next-generation mRNA vaccines and therapeutics.
More description
|

|
| DC67812 | CL15F 6-4 Featured |
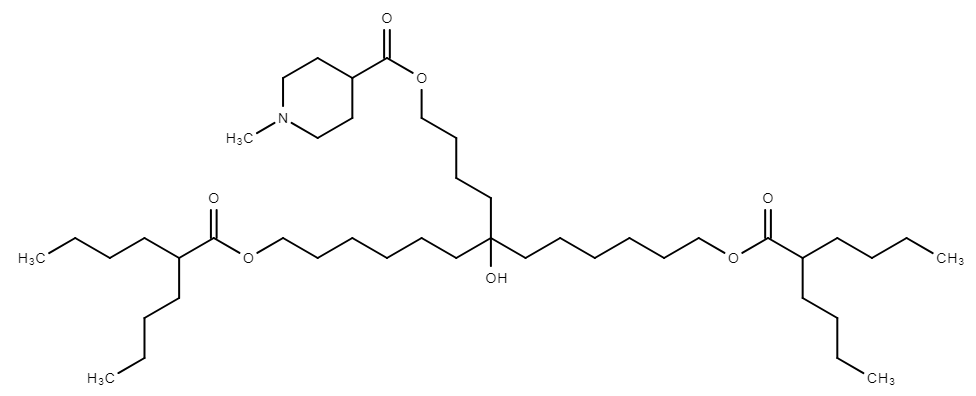
CL15F 6-4 is a short-tail ionizable lipid from the piperidine-based CL15F series, characterized by its symmetric branched structure with a 6-carbon main chain and 4-carbon side chain. This specific tail length critically determines the lipid nanoparticle's (LNP) properties, resulting in larger particles with a high surface density of the phospholipid DSPC. This elevated DSPC density reduces interactions with serum proteins like ApoE, minimizing rapid liver clearance and shifting mRNA delivery preference towards the spleen. Consequently, CL15F 6-4 LNPs achieve efficient, endogenous spleen-targeted delivery, making them a highly promising candidate for enhancing vaccine efficacy by preferentially transfecting antigen-presenting cells without complex functionalization.
More description
|
|
| DC67605 | PyCB lipid Featured |
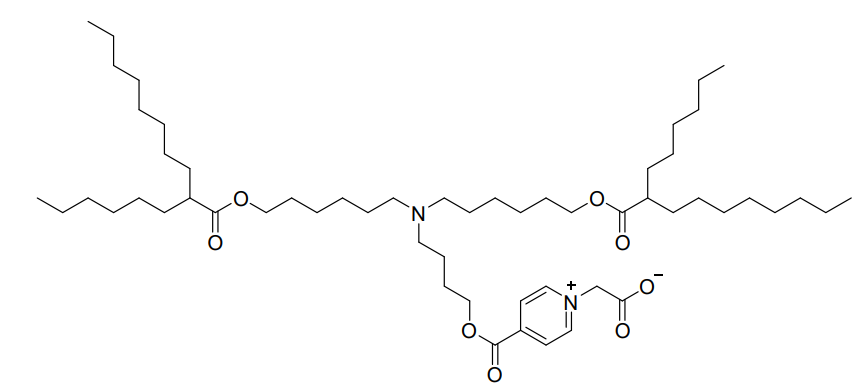
PyCB lipid (MeDZ) is a rationally designed zwitterionic ionizable lipid that serves as a core functional component in the novel three-component (ThrCo) lipid nanoparticle (LNP) platform. It is synthesized by covalently attaching a zwitterionic PyCB structure to the hydroxyl group of the clinically available ionizable lipid ALC-0315.Its key feature is its pH-responsive behavior. At physiological pH (~7.4), the PyCB headgroup exhibits zwitterionic properties, forming charge-assisted hydrogen bonds with water molecules (PyCB-H₂O complexes). This confers high hydrophilicity to the LNP surface, enhancing stability in aqueous environments and reducing nonspecific protein adsorption in the bloodstream. This zwitterionic surface effectively mimics and replaces PEGylated lipids, thereby avoiding PEG immunogenicity and the associated Accelerated Blood Clearance (ABC) effect upon repeated administrations.Crucially, in the acidic environment of endosomes (pH ~6.5), the PyCB group undergoes strong protonation, rapidly transforming into a cationic state (PyCB-H₃O⁺ complexes). This promotes efficient fusion with and disruption of the endosomal membrane, facilitating the escape and cytoplasmic release of encapsulated mRNA.By replacing both cholesterol and PEGylated lipids in traditional LNPs, PyCB lipid enables the redirection of LNP biodistribution from the liver to the spleen, achieving superior spleen-specific mRNA translation and enhancing antigen presentation for potent immune activation.
More description
|
|
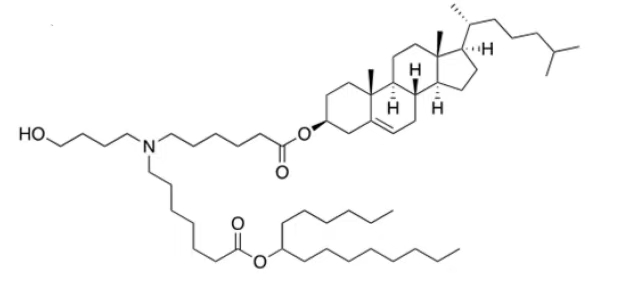
| DC67567 | ARV-T1 Featured |
ARV-T1 is a novel ionizable lipid featuring a cholesterol moiety incorporated in its tail, designed to enhance mRNA delivery efficiency. With a pKa of 6.73, it exhibits optimal pH-dependent ionization for endosomal escape and mRNA release. Structurally, ARV-T1 contains a tertiary amine head group and ester-linked lipid tails, enabling rapid in vivo metabolism and improved biocompatibility.Compared to SM-102 (used in Moderna's vaccine), LNPs formulated with ARV-T1 demonstrate superior physicochemical properties: smaller particle size (~80 nm vs. 90 nm), lower polydispersity index (0.09 vs. 0.10), and higher absolute zeta potential (-10 mV vs. -5 mV). These characteristics correlate with >90% mRNA encapsulation efficiency and enhanced stability, maintaining performance for 12 weeks at -20°C.In vitro, ARV-T1 LNPs showed 7-fold higher protein expression than SM-102 LNPs. In vivo, they prolonged luciferase expression (>72 hours vs. <48 hours for SM-102) and induced 10-fold higher neutralizing antibodies against SARS-CoV-2 spike protein at low doses. The cholesterol tail promotes endosomal membrane fusion, while ester linkages facilitate metabolic clearance, yielding an excellent safety profile in toxicity studies. This combination of efficacy and safety positions ARV-T1 as a promising platform for mRNA vaccines and therapeutics.
More description
|
|
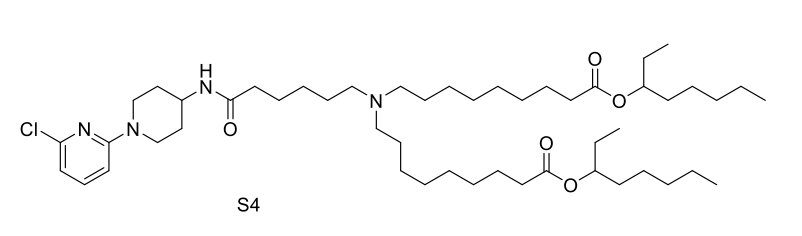
| DC67569 | Lipid S4 Featured |
Lipid S4 is an advanced ionizable lipid engineered for systemic mRNA delivery to the brain, leveraging SR-57227—a high-affinity 5-HT3 receptor ligand—as its core head group to enable targeted blood-brain barrier (BBB) penetration via receptor-mediated transcytosis, while incorporating amino linkers for pH-responsive ionization and biodegradable branched ester tails to facilitate efficient endosomal escape and intracellular mRNA release; optimized through orthogonal screening into OS4 LNP (formulated at S4/DOPE/Chol/DMG-PEG2k = 40:40:60:0.75 molar ratio), it demonstrated a 13.3-fold increase in brain mRNA expression compared to FDA-approved MC3 LNPs, and further conjugation with the Tat cell-penetrating peptide yielded OS4T LNP, boosting delivery efficiency by 12.7-fold over OS4 alone and enabling broad mRNA expression across neurons, astrocytes, microglia, and endothelial cells; validated in orthotopic glioblastoma models, OS4T delivered engineered IL-12 mRNA, suppressing tumor growth and extending median survival to 37 days (vs. 17 days for controls) with minimal systemic toxicity, positioning S4-based LNPs as a robust, translatable platform for CNS-targeted therapeutics.
More description
|
|
| DC67450 | A28-C6B2 Featured |
A28-C6B2 is a biodegradable ionizable lipid specifically engineered for spleen-targeted delivery. Through its unique branched structure, it bypasses hepatic uptake to achieve highly efficient transfection of F4/80+ macrophages and CD11c+ dendritic cells within the spleen. This lipid remains neutral in the bloodstream to minimize non-specific interactions, while undergoing protonation in the acidic environment of the endosome to facilitate nucleic acid release, thereby significantly enhancing the potency of mRNA vaccines and immunotherapies.
More description
|
|
| DC65850 | VL422 Featured |
VL422 is a novel ionizable cationic lipid, as a high-performance "molecular engine" for next-generation Lipid Nanoparticles (LNPs), specifically engineered for the precise delivery of CRISPR base editors and mRNA. Its sophisticated chemical architecture is designed to remain neutral in systemic circulation for enhanced safety, while rapidly protonating within the acidic cellular environment to trigger efficient endosomal escape and cargo release. Validated by groundbreaking research in liver-targeted gene silencing, VL422 has become a critical benchmark molecule for developing permanent, transformative therapies for cardiovascular and metabolic diseases.
More description
|
|
| DC60925 | Lipid H5T5 Featured |
H5T5 is a leading ionizable lipid nanoparticle (LNP) formulation optimized for in vivomRNA delivery, featuring a pKa of 6.51, a size of ~154 nm, and a narrow polydispersity index (PDI) of 0.05. It demonstrated superior in vitromRNA transfection efficiency in primary immune cells, such as bone marrow-derived macrophages. Following intravenous administration, H5T5 exhibits precise organotropism, predominantly targeting the spleen and bone marrow, where it effectively delivers mRNA to a broad spectrum of immune cells, including macrophages, dendritic cells, T cells, B cells, and NK cells. This capability enables its core application: the in vivogeneration of "pan-CAR" immune cells. When loaded with anti-HER2 CAR mRNA, the H5T5-based therapy achieved potent tumor regression and prolonged survival in multiple solid tumor models. Preliminary safety assessments indicated a manageable cytokine profile and no significant organ toxicity, positioning it as a promising platform for in vivocell engineering.
More description
|
|
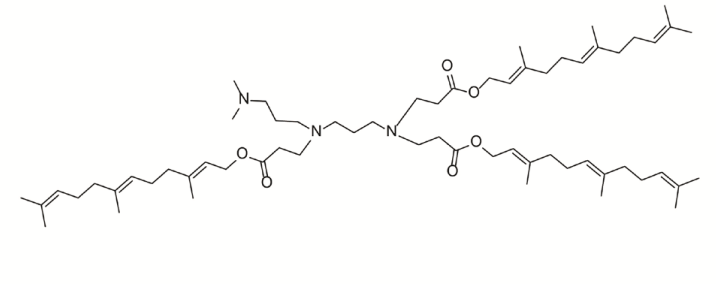
| DC68023 | Lipid 1A7B13 Featured |
1A7B13 is a top-performing "tripod-like" lung-targeting (LuT) lipid. It forms lipid nanoparticles (LNPs) that, after intravenous injection, deliver genetic medicines (like mRNA and CRISPR-Cas9) to the lungs with over 90% selectivity, particularly favoring epithelial cells. Compared to the benchmark DOTAP LNPs, 1A7B13 LNPs achieve a 25.5-fold increase in mRNA delivery and a 9.2-fold improvement in gene-editing efficiency within the lungs. Its therapeutic potential was demonstrated by successfully delivering IL-10 mRNA to treat acute lung injury in mice.
More description
|
|
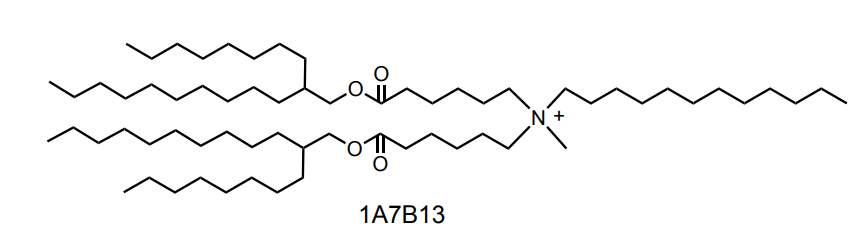
| DC68021 | BIP-20 Featured |
BiP-20 is a branched ionizable phospholipid identified as a lead compound for efficient hepatic mRNA delivery.BiP-20 is a novel, efficient, and safe liver-targeted LNP delivery vehicle. With an ideal pKa of 6.56, it achieves highly efficient liver targeting and endosomal escape primarily through the ApoE/LDL-R pathway. It demonstrates exceptional performance in gene editing at very low doses: for CRISPR-Cas9-mediated editing of TTR, a 10 μg dose achieved ~64% efficiency, which is 8-fold higher than the clinical benchmark lipid LP-01. In Prime Editing targeting the PCSK9 gene, its efficiency (4.30%) also significantly surpassed that of MC3, SM102, and LPO1. Furthermore, it mediates a 5.9-fold increase in hepatic protein expression compared to MC3. Safety assessments indicate it does not induce liver function abnormalities, showing strong therapeutic potential.
More description
|
|
| DC67556 | Sail Lipid 2308 Featured |
Sail Lipid 2308 is a novel ionizable lipid targeting to spleen developed by Sai Biomedicine.As described on US20250205167A1, Lipid 2308 was designed with a piperidine core (6-membered ring) and asymmetric C17/C11 chains, this lipid achieves unprecedented spleen-specificity. It demonstrates dominant spleen accumulation (Spleen RLU: 7.8E+06, 91.8% of total signal) with a record spleen-to-liver ratio of 112.7 (9× higher than 2231). Despite lower protein expression (hEPO: 11,000 ng/mL), near-zero liver uptake (Liver RLU: 66,000) makes Lipid 2308 unparalleled for vaccine/immunotherapy applications targeting splenic immune cells.
More description
|

|
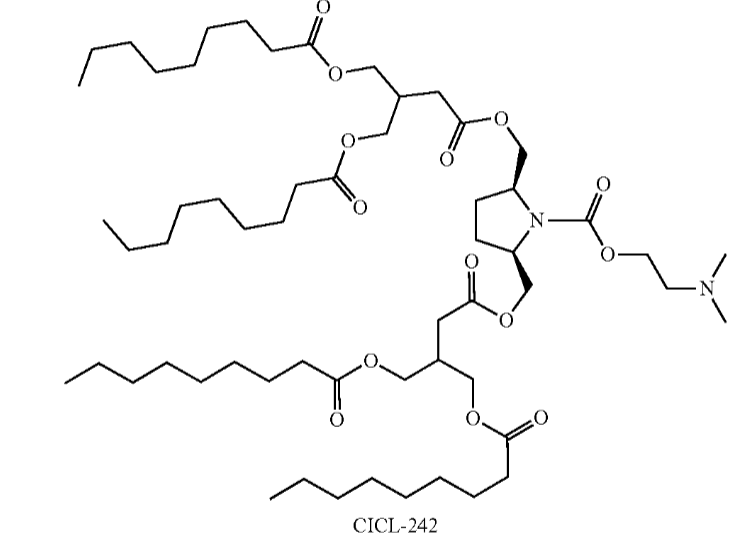
| DC67652 | CICL-242 Featured |
CICL-242 is a constrained ionizable cationic lipid highlighted in patent US 20250127728A1 developed by Capstan as a promising candidate for advanced therapeutic delivery, particularly in stem cell and gene editing applications. Its structure features a rigid amine headgroup similar to CICL-207, which likely facilitates efficient endosomal escape and reduces non-specific uptake, enhancing targeted nucleic acid delivery. Although detailed performance data is not fully disclosed in the patent, CICL-242 is explicitly synthesized and included in gene editing experimental systems (e.g., CRISPR-Cas9 workflows), suggesting its potential for high-efficiency transfection in hard-to-transfect cells like hematopoietic stem cells (CD34⁺). This makes it a strong candidate for ex vivo cell engineering and regenerative medicine, where precision and low off-target effects are critical. While further validation is needed to quantify its efficacy and safety profile, CICL-242 represents a strategic innovation in the lipid library for next-generation genetic therapies.
More description
|
|
| DC68039 | CHCha-10 Featured |
CHCha-10 is an amino acid-derived ionizable lipid engineered for pulmonary gene therapy. Its optimal surface charge (-0.126 mV) enables efficient mucus penetration, while a pKa of ~7.0 facilitates endosomal escape. Its unique conical structure promotes membrane fusion and mRNA release. In animal models, CHCha-10-based LNPs achieved highly efficient and specific editing of lung epithelial cells, particularly stem-like basal cells (~44%), with effects persisting through tissue renewal. It also demonstrated excellent efficacy and safety in ferrets, a model closely mimicking human lung physiology.
More description
|
|
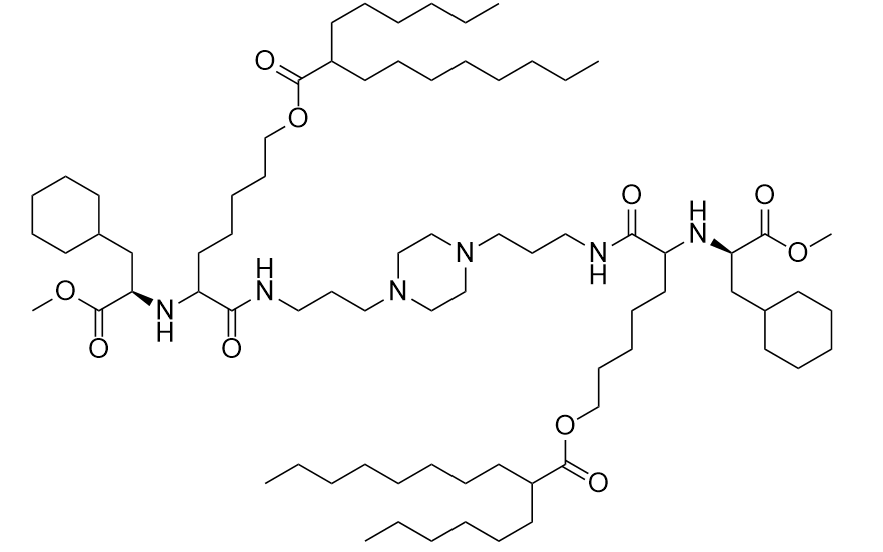
| DC67282 | Merck Lipid X (L608, Merck-32) |
L-608 is a novel ionizable amino lipid designed for formulating lipid nanoparticles (LNPs) to enable efficient subcutaneous (s.c.) delivery of mRNA therapeutics. Engineered to address inflammation associated with mRNA LNPs, L608 integrates seamlessly with steroid prodrugs, such as budesonide-C16 and budesonide-C18:1, to suppress local and systemic inflammatory responses while prolonging therapeutic protein expression. Preclinical studies demonstrate that L608 LNPs significantly reduce injection-site edema (>80% improvement) and lower systemic inflammatory markers (e.g., haptoglobin), while achieving 2–3× higher plasma AUC for proteins like hFGF21 compared to non-steroid LNPs.
More description
|
|
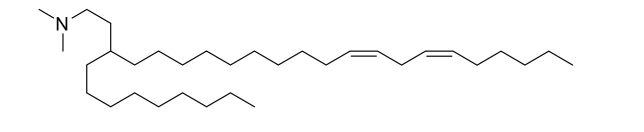
| DC60935 | Lipid D-2 Featured |
D-2 is a novel, custom-synthesized ionizable cationic lipid that serves as the core functional component of the targeted lipid nanoparticle (LNP) delivery system. Its key function is to enable the efficient in vivo delivery of therapeutic mRNAs. Under acidic conditions, it ionizes to a positive charge, allowing it to complex with and encapsulate the negatively charged mRNAs encoding the anti-FAP CAR and Lgmn protease. At physiological pH, it returns to a neutral state, which helps reduce systemic toxicity and is crucial for promoting the release of the mRNA payload inside the target macrophages within the infarcted heart. As part of the optimized LNP formulation, D-2 is fundamental for achieving high transfection efficiency, thereby enabling the in situ generation of efferocytosis-boosted CAR-Ms to treat cardiac fibrosis.
More description
|
|
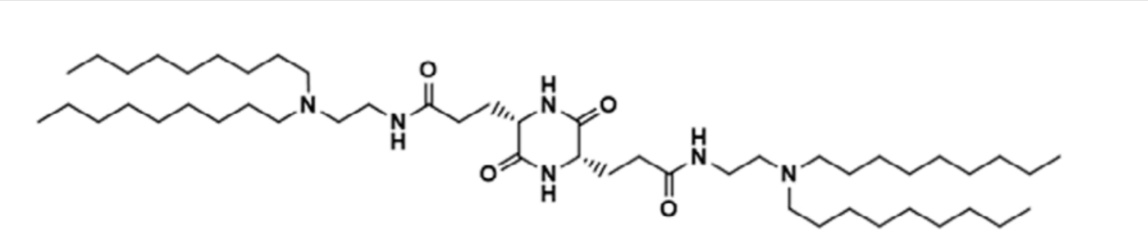
| DC68030 | C6mPhE-383 Featured |
C6mPhE-383 is a top-performing ionizable lipid featuring an aromatic ring and a bioreducible disulfide bond. Formulated into lipid nanoparticles, it preferentially delivers mRNA to lymphoid tissues (lymph nodes/spleen) while minimizing off-target liver accumulation after intramuscular injection. In a SARS-CoV-2 vaccine study, it elicited strong antibody responses, promoted protective effector memory T cells, and exhibited enhanced safety by significantly reducing systemic inflammatory cytokines compared to the standard SM-102 LNP.
More description
|
|
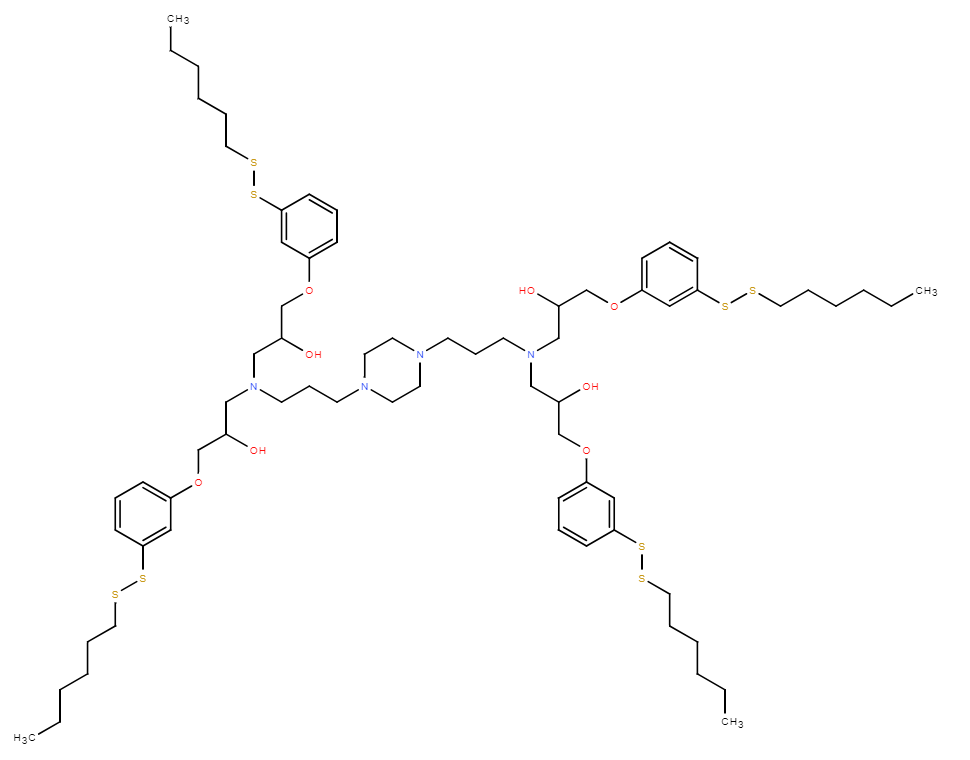
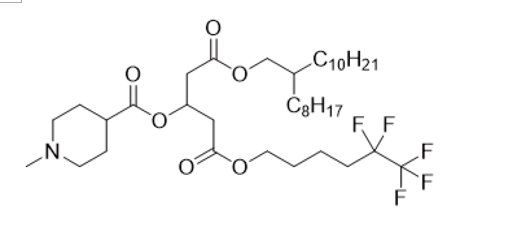
| DC68022 | Lipid A1F5C5 Featured |
A1F5C5 is a core fluorinated ionizable lipid that forms the basis of the F5-LNP platform. Its key biological functions are multifaceted. Primarily, it enables efficient, targeted mRNA delivery in vivo. Following intravenous administration, F5-LNPs exhibit a strong tropism for the spleen and tumor sites, successfully transfecting over 70% of splenic macrophages and more than 20% of tumor-infiltrating macrophages. This allows for in situ cell engineering. Beyond delivery, A1F5C5 possesses intrinsic immunostimulatory activity. It promotes the maturation and activation of antigen-presenting cells (e.g., upregulating CD80/86 on dendritic cells) and enriches immune-related pathways like "cytokine-cytokine receptor interaction." Mechanistically, its unique 5-fluorine (F5) configuration confers superior membrane fusion capability, which is critical for efficient endosomal escape and cytosolic mRNA release. Therapeutically, when loaded with CAR mRNA, it serves as a platform for in vivo generation of CAR-macrophages (CAR-M). These CAR-M cells phagocytose tumors, reprogram the tumor microenvironment by shifting macrophages to an M1 phenotype, and activate CD8+ T cells. Notably, this approach synergizes powerfully with anti-PD-L1 therapy, achieving complete tumor regression in preclinical models.
More description
|
|
| DC60934 | Lipid P3B Featured |
P3B is a biodegradable ionizable lipid engineered to function as a highly efficient delivery vehicle for genome-editing machinery (e.g., CRISPR-Cas9 and adenine base editors) specifically to the central nervous system (CNS). Its primary function is to encapsulate and transport large mRNA payloads across the brain-CSF interface following intrathecal administration, enabling robust and widespread gene editing in neurons and astrocytes across multiple brain regions, including the hippocampus, cortex, and thalamus. Notably, it facilitates precise single-nucleotide correction via base editing. Its targeting is intrinsically achieved by the intrathecal injection route, which localizes the nanoparticles within the cerebrospinal fluid, coupled with an optimized formulation that promotes efficient uptake and activity within CNS parenchyma while minimizing off-target exposure in peripheral organs.
More description
|

|
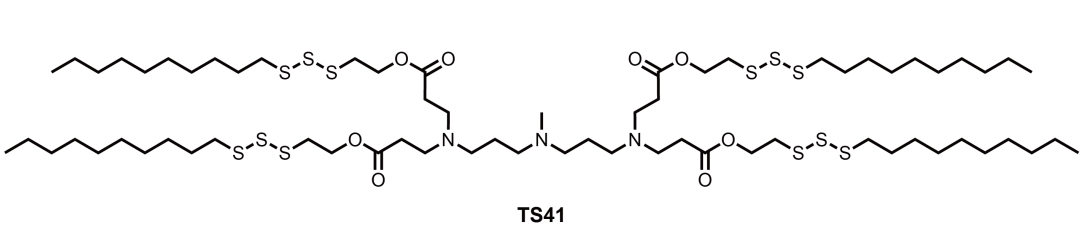
| DC67657 | Lipid TS41 Featured |
TS41 is a trisulfide-derived ionizable lipid engineered for lipid nanoparticles (LNPs) to deliver mRNA therapeutics against multidrug-resistant bacterial pneumonia. Its optimized formulation, TS41S LNP, combines TS41 with helper lipids (e.g., DOPE, cholesterol) at a precise ratio, achieving a hydrodynamic diameter of ~105 nm, low polydispersity, and high mRNA encapsulation efficiency (~84%). This design enables efficient pulmonary delivery via intratracheal administration, with luminescence signals in lungs 4.8-fold higher than clinical benchmarks like SM-102 LNPs, ensuring targeted expression in epithelial cells, macrophages, and neutrophils. Crucially, TS41 LNPs exhibit potent anti-inflammatory properties by scavenging reactive oxygen species (ROS), reducing neutrophil infiltration and proinflammatory cytokines (e.g., IL-6, TNF-α) in infected lungs. In preclinical models, TS41S LNP encoding PB9 peptibody mRNA eradicated pathogens like Staphylococcus aureus and Pseudomonas aeruginosa, improved survival rates to 80%, and minimized tissue damage without systemic toxicity. Its ROS-scavenging capability synergizes with antibacterial effects, offering a promising, translatable platform for combating resistant infections while controlling inflammation. Future enhancements, such as codon optimization or inhalation delivery, could further broaden its therapeutic potential.
More description
|
|
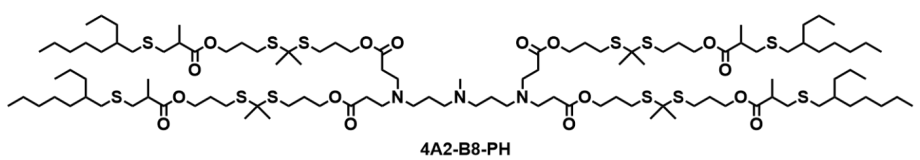
| DC67658 | Lipid 4A2-B8-PH Featured |
4A2-B8-PH is an optimally designed thioketal-incorporated biodegradable ionizable lipid (TBIL) for mRNA delivery to pancreatic ductal epithelial cells. It features a 4A2 headgroup with three tertiary amines, a biodegradable thioketal-based B8 linker, and a branched PH tail. The thioketal linker enables ROS-responsive degradation in the tumor microenvironment, enhancing endosomal escape and mRNA release. In vivo, 4A2-B8-PH LNPs achieve 98.3% pancreas-specific targeting after intraperitoneal administration, with a 218-fold improvement in delivery efficiency compared to previous benchmarks. It successfully transfects 30.5% of pancreatic ductal epithelial cells and induces complete tumor regression in orthotopic PDAC models via IL-12 mRNA therapy, demonstrating high efficacy and safety.
More description
|
|
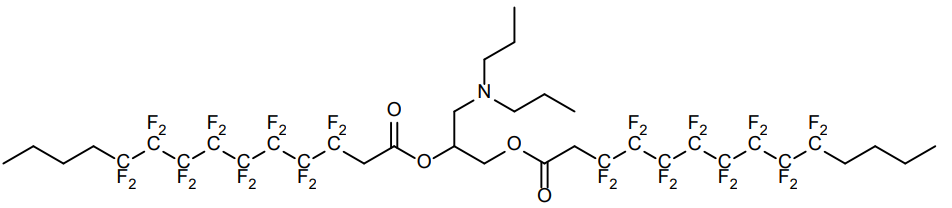
| DC67663 | Lipid 6F Featured |
6F Lipid is a Fluorinated Ionizable Lipid breakthrough in mitochondria-targeted gene delivery
More description
|
|
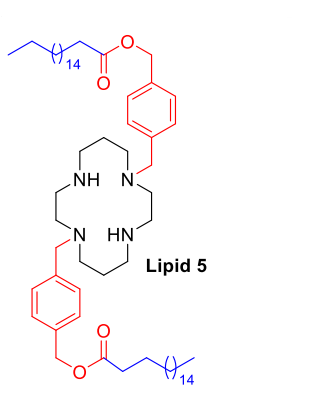
| DC67721 | Macrocyclic Lipid 5 Featured |
Lipid 5 is an ionizable lipid based on a macrocyclic cyclam headgroup. Its structure incorporates a benzylmethyl carbonate (BMC) linker, which contains an aromatic benzene ring, and a saturated C18 hydrophobic tail. Lipid 5 was mixed with helper lipids at a fixed molar ratio and formulated into mRNA-loaded lipid nanoparticles (LNPs) using microfluidic technology. Characterization data show that these LNPs have a hydrodynamic diameter of approximately 50-80 nanometers and a polydispersity index (PDI) below 0.2, indicating a small particle size with a uniform distribution. Their zeta potential at physiological pH is near neutral (ranging from -3 to +3 mV). The mRNA encapsulation efficiency, as determined by the Ribogreen assay, exceeds 95%. Cryo-transmission electron microscopy images reveal that the LNPs exhibit a typical spherical bilayer structure. In in vitro experiments, Lipid 5 LNPs mediated a higher level of luciferase protein expression in HEK293FT cells compared to the benchmark lipid DLin-MC3-DMA. In Balb/c mice, intravenous injection of LNPs encapsulating luciferase mRNA resulted in in vivo imaging signals predominantly concentrated in the lungs. Quantitative analysis indicated that the signal intensity in the lungs was over 100 times greater than that in the liver, with more than 95% of the total signal distributed in the lungs. In Ai9 reporter gene mice, two intravenous injections of Lipid 5 LNPs encapsulating Cre mRNA led to quantitative analysis of lung tissue sections showing that approximately 30% of lung cells were positive for tdTomato signal.
More description
|
|
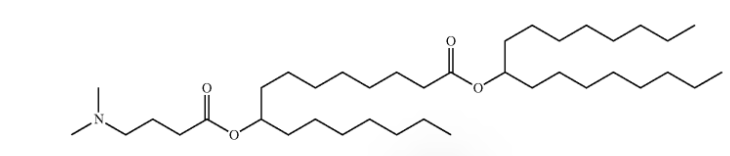
| DC67570 | Generation Lipid 87 Featured |
Lipid-87 is an ionizable lipid developed by Generation Bio, characterized by its tertiary amine group for pH-dependent protonation and dual C16/C17 aliphatic chains that enhance hydrophobic stability.As the core component (47.5–57.5 mol%) of stealth lipid nanoparticles (LNPs), Lipid87 enables extended blood circulation (>24-hour half-life vs. 30 min for conventional LNPs) by synergizing with steric-stabilizing polymers (e.g., DSG-PEG₂₀₀₀-OMe), achieves >95% encapsulation efficiency for mRNA/ceDNA with low cytotoxicity (IC₅₀ >100 μM), and drives liver-specific targeting (>80% hepatocyte transfection at 0.5 mpk), effectively restoring 40% FIX activity in hemophilia B models for over 7 days.
More description
|
|
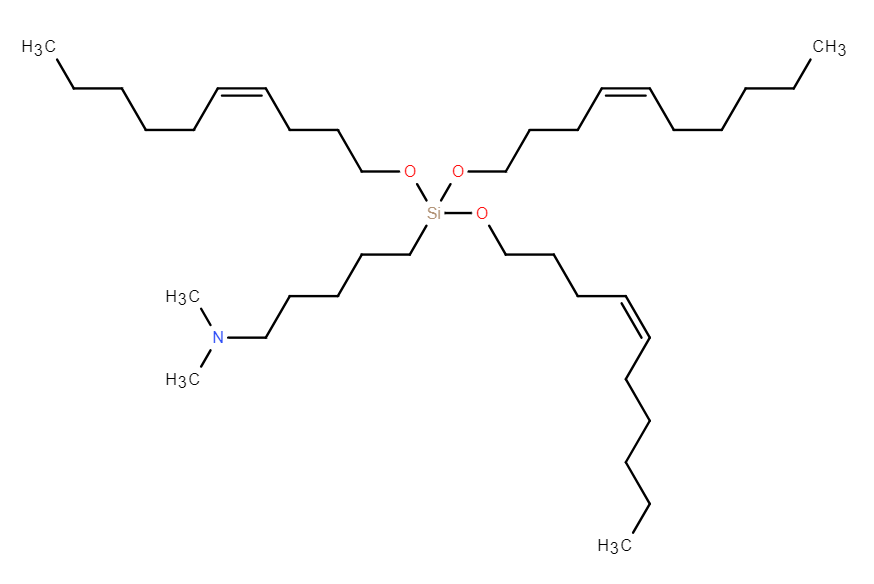
| DC67616 | GVS-18-B34 |
GVS-18-B34 is a highly potent, silicon ether-based ionizable lipid that enables efficient mRNA delivery via lipid nanoparticles (LNPs). Its key advantage lies in a biodegradable silyl ether linkage, which undergoes rapid, non-enzymatic hydrolysis, leading to near-complete clearance from the liver within 24 hours in both mice and non-human primates (NHPs). This degradation mechanism is species-agnostic, overcoming the variability associated with esterase-dependent lipids. In vivo, GVS-18-B34 LNPs demonstrated superior liver-specific protein expression and a high liver-to-spleen signal ratio, indicating minimal off-target accumulation and reduced immune stimulation compared to benchmarks like MC3 and SM-102. The LNPs exhibited excellent stability when stored frozen at -80°C, maintaining integrity over multiple freeze-thaw cycles. With its optimal pKa (~6.3) and efficient endosomal escape profile, GVS-18-B34 represents a promising candidate for therapeutic applications requiring frequent dosing, combining high potency with a favorable safety profile derived from its rapid clearance.
More description
|
|
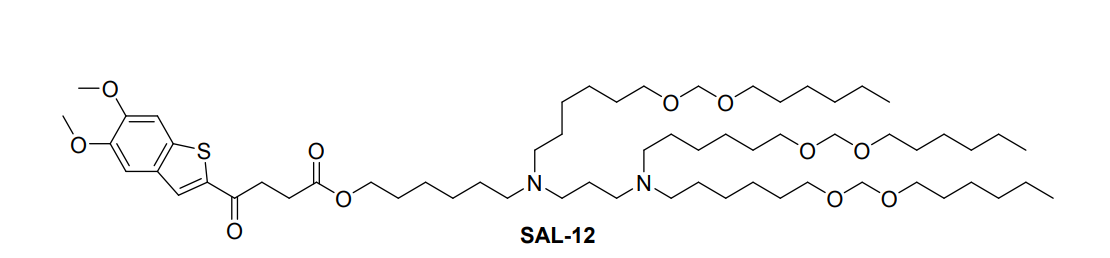
| DC67615 | STING Agonist Lipid SAL-12 |
SAL12 is a novel ionizable lipid derivative that integrates a non-nucleotide STING agonist (agonist 6) with an amino lipid tail through an ester bond, forming the core component of specialized lipid nanoparticles (SAL12-LNPs). These nanoparticles are designed for dual functionality: they efficiently encapsulate and deliver mRNA into dendritic cells while concurrently activating the STING pathway to stimulate innate immunity.
More description
|
|
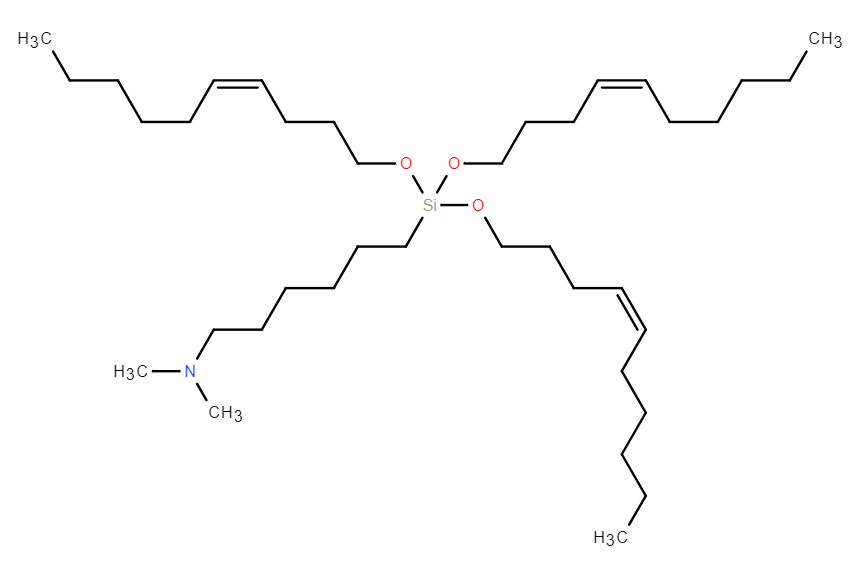
| DC67618 | GVS-18-B35 |
GVS-18-B35 is a leading silicon ether-based ionizable lipid that demonstrates exceptional performance in mRNA delivery. It features a biodegradable silyl ether linkage, which undergoes rapid, non-enzymatic hydrolysis, enabling near-complete clearance from the liver within 24 hours in both mice and non-human primates (NHPs). This degradation mechanism is independent of variable enzymatic activity, ensuring consistent pharmacokinetics across species. In vivo, GVS-18-B35 lipid nanoparticles (LNPs) achieve superior liver-specific protein expression with minimal off-target accumulation in the spleen, resulting in a high liver-to-spleen signal ratio and reduced immune stimulation. The LNPs exhibit excellent stability under frozen storage (-80°C) and maintain critical quality attributes, including particle size, polydispersity, and encapsulation efficiency, through multiple freeze-thaw cycles. With an optimal pKa (~6.3) and enhanced endosomal escape capability, GVS-18-B35 represents a robust and versatile platform for mRNA therapeutics, particularly suited for applications requiring frequent dosing due to its unique combination of high potency and rapid clearance profile.
More description
|
|
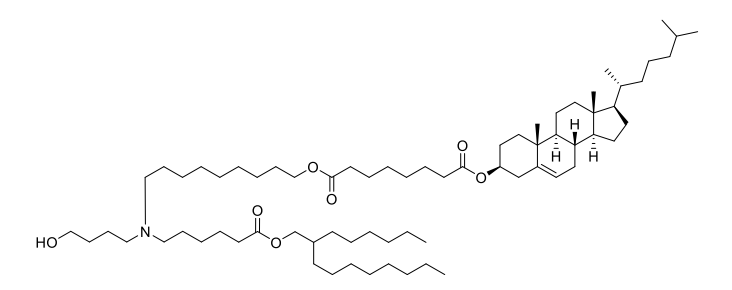
| DC67617 | iChol15-C4A2 |
iChol15-C4A2 is a groundbreaking ionizable cholesteryl lipid, expertly designed to overcome the primary challenge of liver-centric accumulation in mRNA therapeutics. Its innovative "two-in-one" structure seamlessly integrates cholesterol with an ionizable headgroup, enabling the formation of stable, three-component Lipid Nanoparticles (Tc-LNPs).The key advantage of Tc-LNPs formulated with iChol15-CA2 is their significantly reduced adsorption of Apolipoprotein E (ApoE).This unique property directly attenuates ApoE/LDLR-mediated uptake by liver cells, dramatically shifting biodistribution toward extrahepatic tissues. Peer-validated research demonstrates a remarkable 20-50 fold increase in the spleen-to-liver mRNA expression ratio compared to standard LNPs like ALC-0315, unlocking unparalleled potential for targeting the immune system.
Beyond its superior targeting capability, iChol15-C4A2 ensures high mRNA encapsulation efficiency, excellent colloidal stability, and proven biocompatibility. It offers a powerful, off-the-shelf solution to advance next-generation mRNA applications, from innovative vaccines and cancer immunotherapies to treatments for splenic disorders. Discover how iChol15-C4A2 can transform your delivery platform.
More description
|
|
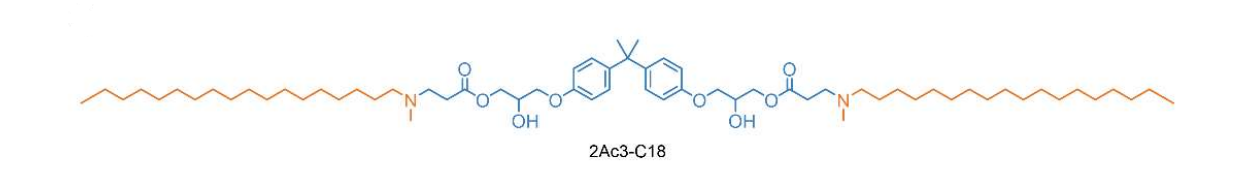
| DC60880 | 2Ac3-C18 Featured |
2Ac3-C18 is a unique ionizable lipid with a distinct degradable core structure:featuring 2 acrylate units and 3 amine groups—linked to a C18 alkyl chain. Its LNPs (formulated with DOPE/cholesterol/DMG-PEG2000) exhibit spleen-specific mRNA delivery in vivo.
More description
|
|