 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
| Cat. No. | Product Name | Field of Application | Chemical Structure |
|---|---|---|---|
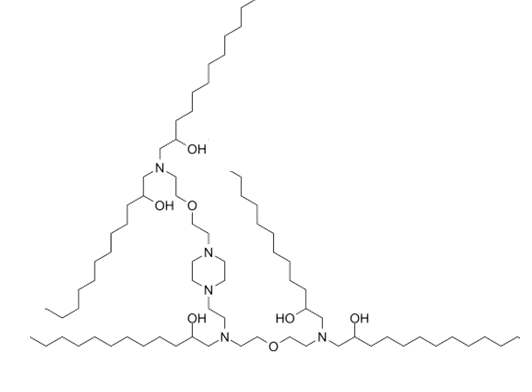
| DC89101 | C12-4 (Lipid A-4,Lipid F3) Featured |
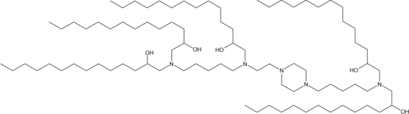
C12-4 (Lipid A-4,Lipid F3) is a branched-chain ionizable cationic lipidoid that has been used in the formation of lipid nanoparticles (LNPs) for the delivery of mRNA. LNPs containing lipid A4 and encapsulating an mRNA reporter accumulate in the uterus, placenta, and ovaries, as well as to the spleen and liver, in pregnant mouse dams unlike LNPs containing the branched-chain ionizable cationic lipidoid C12-200, which primarily accumulate in the liver. Intravenous administration of LNPs containing lipid A4 and encapsulating mRNA encoding VEGF increase placental VEGFR1 levels and mean fetal blood vessel area without inducing liver damage in pregnant mouse dams.
More description
|
|
| DC60839 | A2T2C9 (CP-LC-1465) Featured |
A2T2C9 (CP-LC-1465) is an imidazole-based ionizable lipid with branched hydrophobic chains and β-propionate linkers, formulated in four-component LNPs for spleen-targeted mRNA delivery. It achieves >80% spleen selectivity with protein expression exceeding 1×10⁷ p/s in mice, driven by its negative zeta potential (-9.7 to -19 mV). Structural features including imidazole polar head and branched acrylate (C9) enhance splenic tropism, potentially through distinct protein corona interactions. Demonstrated low cytotoxicity (>75% viability in splenic cells) and biodegradability via pH-sensitive linkers enable efficient mRNA delivery without permanent charged additives, outperforming conventional anionic SORT systems in selectivity and therapeutic potential.
More description
|
.png)
|
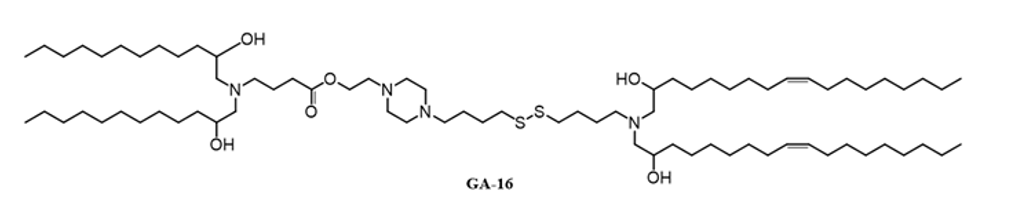
| DC68058 | Lipid GA-16 Featured |
GA-16 is a novel ionizable lipid designed with an N-(2-Hydroxyethyl)piperazine-N'-(4-butanesulfonic acid) (HEPBS) core. Its key functional characteristic is enabling highly efficient, liver-tropic mRNA delivery through an ApoE-mediated yet Low-Density Lipoprotein Receptor (LDLR)-independent cellular uptake mechanism. When formulated in LNPs with the helper lipid DEPE, GA-16 achieves superior protein expression. More importantly, this uptake pathway bypasses the canonical LDLR dependency, making it a strategically valuable component for developing therapies targeting genetic liver diseases like familial hypercholesterolemia, where patients often have deficient LDLR function.
More description
|
|
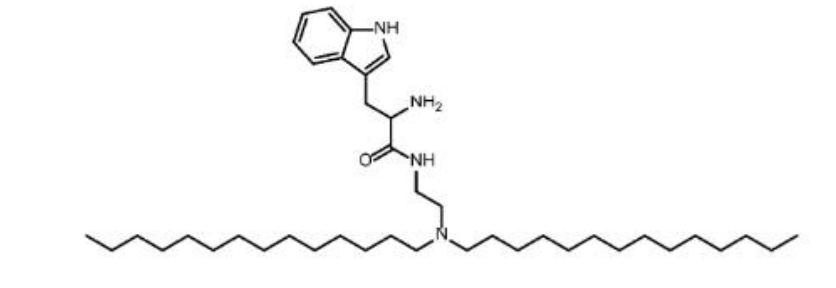
| DC68057 | lipid Trp-L1-T4 Featured |
Trp-L1-T4 is a novel tryptophan-derived ionizable lipid that serves as the core functional component of the optimized lipid nanoparticle (TLNP/RLNP) platform. Its primary function is to enable the efficient encapsulation and in vivo delivery of self-amplifying RNA (saRNA) cargo. Specifically, it facilitates high transfection efficiency and cytosolic release of the RNA payload in target follicular helper T (Tfh) cells, with minimal cytotoxicity. This capability is crucial for reprogramming pathogenic Tfh cells into regulatory CAR-Tfh cells, forming the foundation of the study's therapeutic strategy.
More description
|
|
| DC68056 | A1T2C3(CP-LC-1067) Featured |
A1T2C3 (CP-LC-1067) is a highly potent ionizable lipid designed for lung-targeted mRNA delivery. When formulated into lipid nanoparticles (LNPs), it produces particles approximately 80 nm in size with excellent uniformity and a high mRNA encapsulation efficiency (>94%). Its most distinguished feature is its exceptional organ selectivity. Following intravenous administration in mice, A1T2C3-based LNPs demonstrated a remarkable accumulation of over 94% of the signal in the lungs, with minimal distribution to other major organs. This makes it a leading candidate for developing therapeutics specifically targeted to pulmonary tissues.
More description
|
.gif)
|
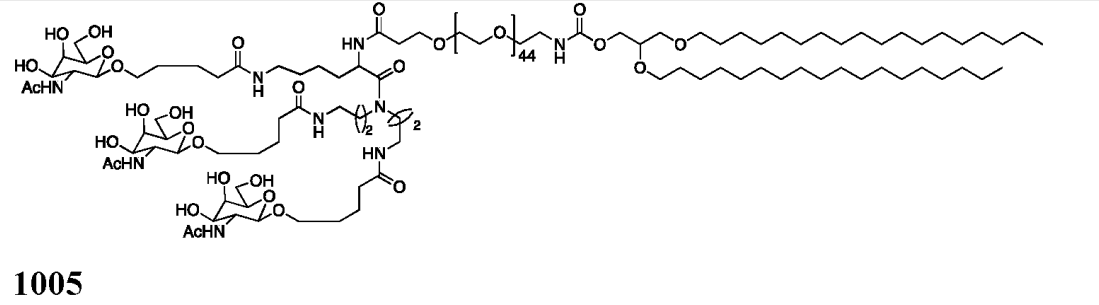
| DC67444 | GalNAc Lipid 1005 Featured |
GalNAc Lipid 1005 is a trivalent GalNAc-lipid conjugate designed for ASGPR-mediated hepatic delivery. It features a lysine-based scaffold covalently linked to three GalNAc moieties via a 44-unit PEG spacer, anchored by a 1,2-O-dioctadecyl-sn-glyceryl (DSG) lipid tail.
More description
|
|
| DC60502 | GalNAc Lipid GL6(GalNAc Lipid 1004) Featured |
GL6 is a trivalent GalNAc-lipid conjugate designed for ASGPR-mediated hepatic delivery. It features a lysine-based scaffold covalently linked to three GalNAc moieties via a 36-unit PEG spacer, anchored by a 1,2-O-dioctadecyl-sn-glyceryl (DSG) lipid tail. This structure balances ligand accessibility (via optimized PEG length) and nanoparticle stability (via hydrophobic DSG anchoring). Compared to GL3 (TRIS scaffold, same PEG length), GL6’s simplified lysine scaffold improves manufacturability. In LDLR-deficient models, GL6 enabled 61% liver editing (vs. 5% with standard LNPs) at 2 mg/kg, demonstrating superior ASGPR targeting. Its design minimizes ligand crowding (0.05 mol% surface density) while maximizing endosomal escape and durable gene editing.
More description
|

|
| DC67443 | GalNAc Lipid 1002 Featured |
GalNAc Lipid 1002 is a trivalent GalNAc-lipid conjugate designed for ASGPR-mediated hepatic delivery. It features a lysine-based scaffold covalently linked to three GalNAc moieties via a 12-unit PEG spacer, anchored by a 1,2-O-dioctadecyl-sn-glyceryl (DSG) lipid tail.
More description
|
|
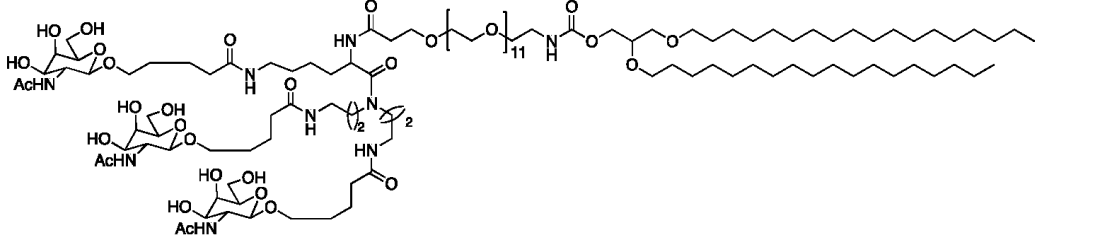
| DC67408 | Galnac Lipid 29 Featured |

|
|
| DC67409 | Galnac Lipid 83 Featured |
Galnac Lipid 83 is developed by Prime Medicine Patent: WO2024220807.Galnac Lipid 83 83 is a GalNAc-conjugated lipid designed for targeted liver delivery. It features a triantennary GalNAc ligand linked via a PEG spacer (e.g., -(CH2CH2O)n-) to a branched hydrophobic tail (C18 alkyl chains). The structure includes amide/ester bonds for stability and a stereospecific configuration (R/S) to optimize ASGPR receptor binding. Integrated into lipid nanoparticles (LNPs), it enhances hepatic uptake of nucleic acids (e.g., mRNA, gene editors) by leveraging ASGPR-mediated endocytosis. Its design balances hydrophilicity (PEG) and lipophilicity (alkyl chains) for efficient encapsulation and in vivo delivery, supporting therapeutic applications in liver-specific gene editing or RNA therapies.
More description
|

|
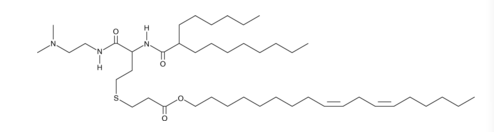
| DC66648 | CP-LC-1143 |
Lipid CP-LC-1143 is an ionizable cationic amino lipid derived from homocysteine, a naturally occurring amino acid. This lipid has demonstrated an efficient delivery and high protein expression of different kinds of RNA (mRNA, cRNA and saRNA) in vivo, with no signs of toxicity.
More description
|
|
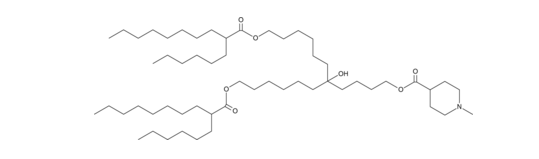
| DC65851 | CL15F6 |
CL15F6 is an ionizable cationic lipid (pKa = 6.75).1 It has been used in the formation of lipid nanoparticles (LNPs) for the delivery of mRNA and polymer-lipid hybrid nanoparticles for the delivery of plasmid DNA in vitro.1,2
More description
|
|
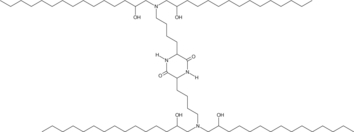
| DC65620 | cKK-E15 |
cKK-E15 is an ionizable cationic lipid and a derivative of cKK-E12 that has been used in the generation of lipid nanoparticles (LNPs).1 LNPs containing cKK-E15 and encapsulating Cre mRNA induce the expression of Cre in Kupffer cells, endothelial cells, and hepatocytes in Ai14 mice engineered to express the fluorescent protein tdTomato upon translation of Cre.
More description
|
|
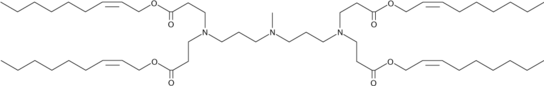
| DC65622 | 306Oi9-cis2 |
306Oi9-cis2 is an ionizable cationic lipid.
WARNING This product is not for human or veterinary use.
More description
|
|
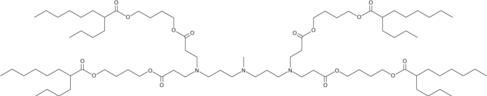
| DC65623 | Lipid AX4 |
Lipid AX4 is an ionizable cationic lipid (pKa = 6.89) that has been used in the formation of lipid nanoparticles (LNPs) for the delivery of mRNA in vivo.
More description
|
|
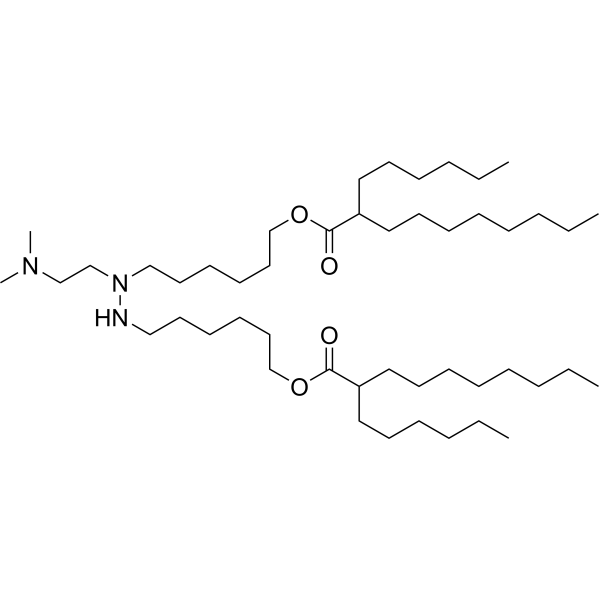
| DC65439 | LNP Lipid-6 |
LNP Lipid-6 (Compound Lipid 5) is an ionizable lipid (amino lipid). LNP Lipid-6 can be used to prepare lipid nanoparticles (LNP).
More description
|
|
| DC65437 | LNP Lipid-2 |
LNP Lipid-2 is a lipid product can be used to deliver agents.
More description
|
|
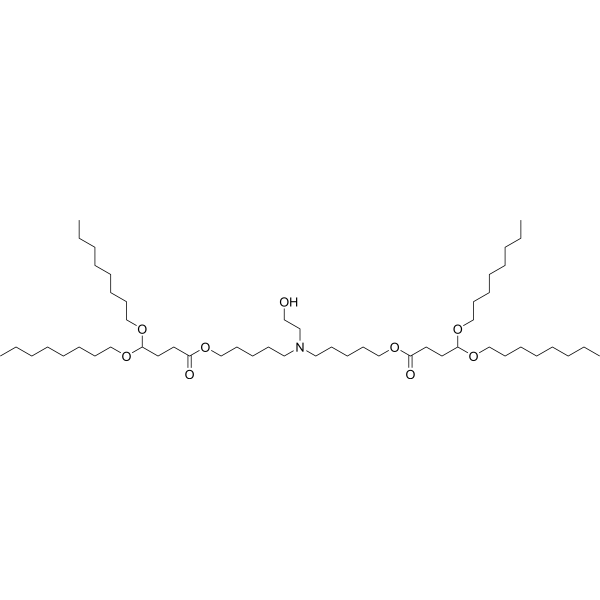
| DC65438 | SM-102 Analog 2(Compound 8-8) |
SM-102 Analog 2(Compound 8-8) is a lipid compound. SM-102 Analog 2(Compound 8-8) is involved in the synthesis of lipid nanoparticles compositions. SM-102 Analog 2(Compound 8-8) has potential applications in the transportation of biologically active substances.
More description
|
|
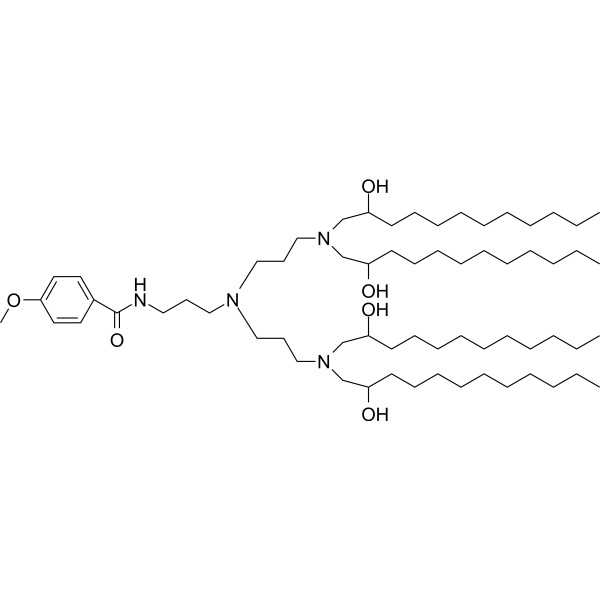
| DC65328 | AA-T3A-C12 Featured |
AA-T3A-C12 is a leading anisamide-tethered lipidoid (AA-lipidoid) identified through a combinatorial library screening for targeted RNA delivery to activated fibroblasts, offering a promising approach to treat liver fibrosis.AA-T3A-C12 is a leading anisamide-tethered lipidoid (AA-lipidoid) identified through a combinatorial library screening for targeted RNA delivery to activated fibroblasts, offering a promising approach to treat liver fibrosis. It is synthesized via a one-pot, two-step modular method that combines anisamide—a ligand for sigma receptors overexpressed on activated hepatic stellate cells (HSCs)—with a T3A polyamine core and C12 epoxide tails, enabling efficient siRNA encapsulation in lipid nanoparticles (LNPs).
In vitro, AA-T3A-C12 LNPs exhibit enhanced cellular uptake and gene silencing in activated fibroblasts, dependent on sigma receptor binding, as confirmed by haloperidol blockade studies, and outperform non-targeted analogs and the FDA-approved MC3 LNPs in fibroblast selectivity.In a mouse model of CCl4-induced liver fibrosis, AA-T3A-C12/siHSP47 LNP achieves approximately 65% knockdown of heat shock protein 47 (HSP47), a key fibrotic target, leading to significant reduction in collagen deposition and fibrosis alleviation, with a good safety profile and no exacerbation of liver injury.
More description
|
|
| DC60215 | Moderna Lipid 29 Featured |
Lipid 29 is an ionizable amino lipid (pKa = 6.91) from Moderna platform that has been used in combination with other lipids in the formation of lipid nanoparticles (LNPs).Administration of human erythropoietin (EPO) mRNA in lipid 29-containing LNPs increases serum EPO levels in mice.
More description
|

|
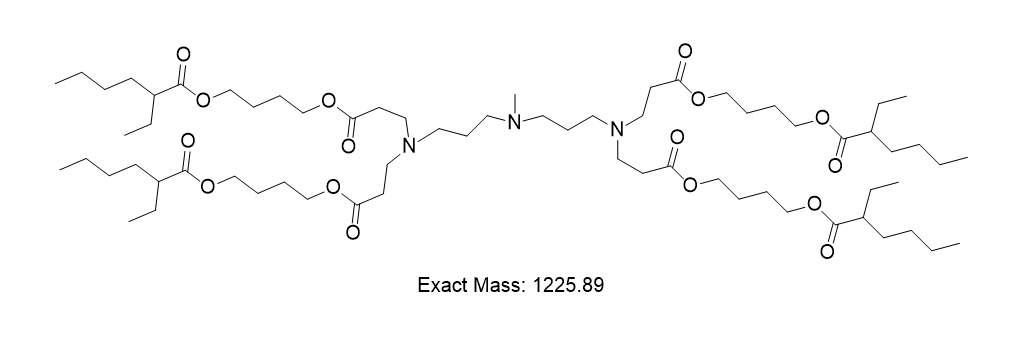
| DC60579 | Lipid B3 Featured |
Lipid B3 is a biodegradable ionizable lipid for liver targeted delivery. Lipid B3-LNP shows high delivery efficacy and low toxicity in delivering RNA to liver cells.
More description
|
|
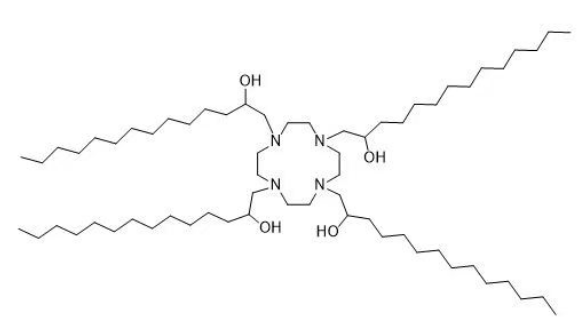
| DC60842 | Lipid H7T4-4 Featured |
H7T4-4 is an ionizable lipid designed for mRNA delivery via lipid nanoparticles (LNPs). It features a cyclic amine headgroup (derived from cyclen tetrahydrochloride) and four C14 hydrophobic alkyl tails, synthesized through a Michael addition reaction between cyclen and 1,2-epoxytetradecane. With a high transition temperature (Tm = 58.6°C) due to strong intermolecular interactions from its cyclic headgroup and multi-tail structure, H7T4-4 alone forms rigid aggregates incompatible with mRNA encapsulation. However, when blended with low-Tm helper lipids (e.g., DOPE, Tm = -16°C), the system’s overall Tm decreases, enabling stable LNP formation. Optimized formulations (20% H7T4-4, 41% DOPE, 38% cholesterol, 1% DMG-PEG) exhibit efficient mRNA encapsulation (>90%) and transfection. Structural analyses (SAXS, cryo-TEM) confirm monodisperse LNPs with lamellar/hexagonal phases. In vivo, H7T4-4 LNPs show tumor-targeted and intranasal mRNA delivery with reduced off-target accumulation compared to SM-102-based LNPs. This rational design highlights Tm-guided helper lipid selection to overcome rigidity challenges in ionizable lipids.
More description
|
|
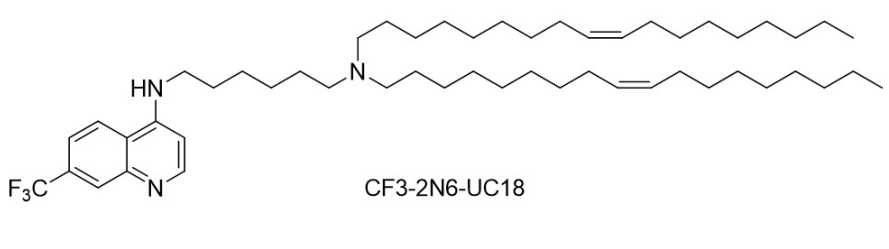
| DC60843 | CF3-2N6-UC18 Featured |
CF3-2N6-UC18 is a rationally designed chloroquine-inspired ionizable lipid that enables robust mRNA delivery and genome editing. It integrates three modular components: a 7-trifluoromethyl-substituted quinoline scaffold (mimicking chloroquine’s endosomolytic properties), a hexamethylenediamine linker with two ionizable nitrogen atoms (pH-responsive protonation), and two unsaturated oleyl (C18:1) hydrophobic tails (enhancing membrane fusion and nanoparticle stability). This lipid self-assembles into ecoLNPs (endosomolytic chloroquine-like lipid nanoparticles) with spherical morphology (~200 nm diameter, 98% mRNA encapsulation). Its pH-sensitive activity triggers endosomal escape through dual mechanisms: proton sponge effect (buffering endo-lysosomal pH) and saposin B-mediated membrane disruption (molecular docking confirms chloroquine-like binding to lysosomal saposin B). In vitro, ecoLNPs outperform commercial reagents (18.9-fold higher mRNA delivery than Lipofectamine 2000) and penetrate 3D cell models. They resist serum/RNase degradation and retain >90% activity after 7-day storage at 4°C. In vivo, ecoLNPs achieve tissue-specific mRNA expression via multiple routes (intravenous, intramuscular, etc.), with strong lymph node tropism (90.2% after intramuscular injection) comparable to SM-102 LNPs (Moderna’s COVID-19 vaccine carrier). They mediate efficient Cre mRNA-driven recombination and CRISPR-Cas9 editing in transgenic mice. CF3-2N6-UC18’s modular design, stability, and dual endosomal escape strategies position it as a versatile platform for mRNA vaccines, gene therapy, and genome editing applications.
More description
|
|
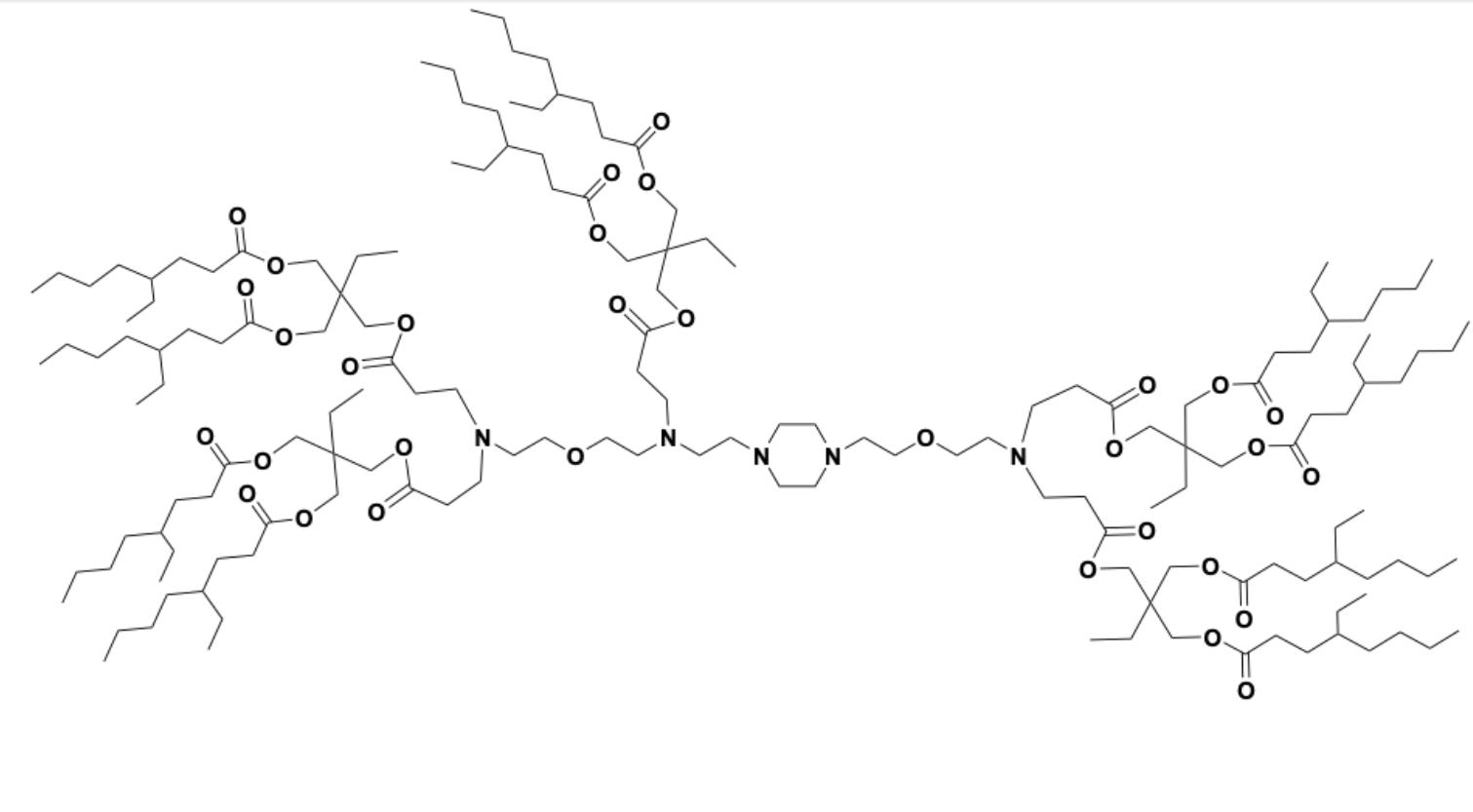
| DC60800 | 18-2-9b2 Featured |
18-2-9b2 is a dendron-like degradable ionizable lipid which facilitates mRNA delivery to splenic macrophages. 18-2-9b2 LNP encapsulating therapeutic BTB domain and CNC homologue 1 (BACH1) mRNA exhibited proficient BACH1 expression and subsequent Spic downregulation in splenic red pulp macrophages (RPM) in a Spic-GFP transgene model.
More description
|
|
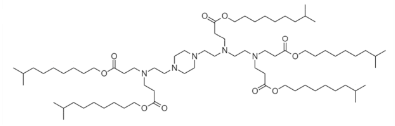
| DC60545 | 200Oi10 Featured |
200Oi10 is an ionizable lipidoid used in lipid nanoparticles (LNPs) for RNA delivery. Structurally, it features ester-conjugated cleavable lipid tails, enhancing biodegradability and reducing toxicity compared to non-cleavable analogs. Preclinical studies show that 200Oi10-based LNPs primarily accumulate in the liver (97.7%) after intravenous administration. However, intraperitoneal injection redirects biodistribution, achieving 46.4% pancreatic uptake, which can be further amplified by incorporating cationic lipids like DOTAP. This unique tropism enables pancreas-targeted mRNA delivery. 200Oi10's ester linkages promote rapid clearance, improving biocompatibility while maintaining siRNA/mRNA delivery efficiency. Its design exemplifies the use of degradable lipidoids to balance organ specificity, efficacy, and safety in RNA therapeutics.
More description
|
|
| DC41043 | NT1-O12B Featured |
NT1-O12B, an endogenous chemical and a neurotransmitter-derived lipidoid (NT-lipidoid), is an effective carrier for enhanced brain delivery of several blood-brain barrier (BBB)-impermeable cargos. Doping NT1-O12B into BBB-impermeable lipid nanoparticles (LNPs) gives the LNPs the ability to cross the BBB. NT-lipidoids formulation not only facilitate cargo crossing of the BBB, but also delivery of the cargo into neuronal cells for functional gene silencing or gene recombination.
More description
|

|
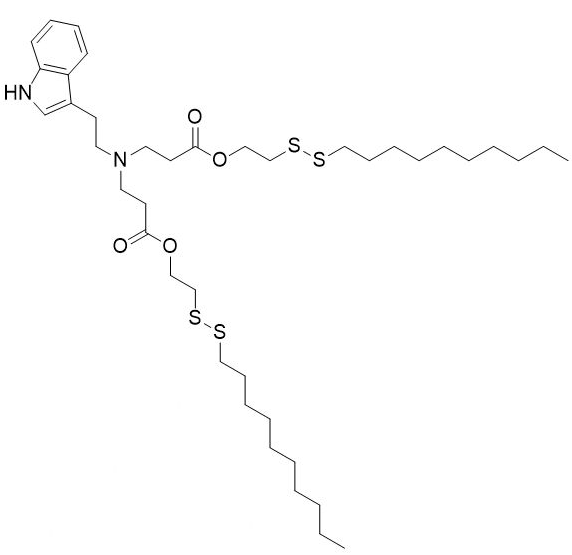
| DC60212 | NT1-O14B Featured |
NT1-O14B is a tryptamine-containing cationic lipidoid.1 It has been used in combination with other lipids in the formation of lipid nanoparticles (LNPs). Intravenous administration of LNPs containing NT1-O14B and encapsulating antisense nucleotides against tau decreases tau brain levels in mice.
More description
|
|
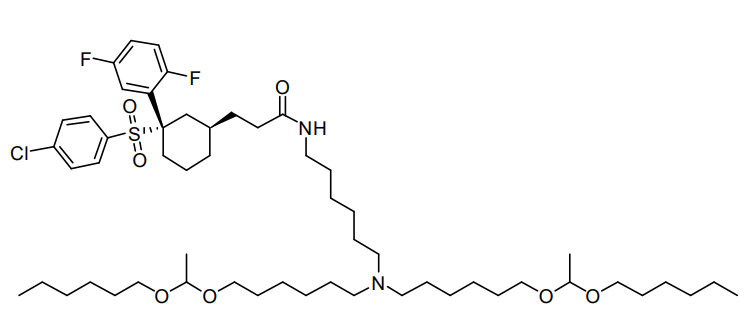
| DC67295 | Lipid MK16 Featured |
MK-16 is a specialized lipid designed to traverse the blood-brain barrier (BBB) for effective mRNA delivery. Its formulation, MK 16 BLNP, leverages dual mechanisms involving caveolae and γ-secretase to facilitate BBB penetration, ensuring the targeted and efficient transport of functional mRNA to diverse brain cell types. Demonstrating excellent tolerability across a range of dosing regimens, MK16 BLNP represents a promising platform for brain-targeted therapeutic applications.
More description
|
|
| DC67521 | Lipid TD5 Featured |
TD5 is a brain-targeting lipid nanoparticle (BLNP) engineered for efficient mRNA delivery to the central nervous system (CNS) via intrathecal injection. It incorporates a tryptamine-derived ionizable lipid headgroup, myristic acid hydrocarbon tails, and a biodegradable carbonate ester linker, enabling pH-dependent mRNA encapsulation (81.7% efficiency) and brain cell-specific targeting. With a hydrodynamic diameter of 107.5 nm, near-neutral pKa (7.30), and mild positive charge, TD 5 demonstrates superior CNS tropism through serotonin receptor (5-HT1A)-mediated endocytosis. In vitro, TD-5 achieved 80.8% GFP expression in SH-SY5Y neuronal cells, outperforming MC3 LNPs by 50-fold. Following intrathecal administration in mice, TD-5 mediated GFP expression in 29.6% of neurons and 38.1% of astrocytes brain-wide, with 10-fold higher CNS specificity than peripheral organs. Genome editing studies showed TD5-delivered Cas9/sgRNA induced tdTomato activation in ≈30% of neurons and 40% of astrocytes across key brain regions. Safety profiling revealed minimal systemic immune responses (lower IL-6, IL-12p40 vs MC3 LNPs), normal hepatic/renal biomarkers, and no histopathological toxicity. The optimized structure balances myristic chain hydrophobicity for membrane interaction, ionizable amines for mRNA complexation, and tryptamine-mediated targeting for enhanced CNS uptake, establishing TD5 as a promising platform for CNS gene therapies.
More description
|

|
| DC60499 | C14-A1 Featured |
Lipid C14-A1 is an ionizable lipid. C14-A1-LPN is a potent and safe LNP platform to deliver Foxp3 mRNA to CD4+ T cells to engineer immunosuppressive FP3T cells.
More description
|
|