 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
| Cat. No. | Product Name | Field of Application | Chemical Structure |
|---|---|---|---|
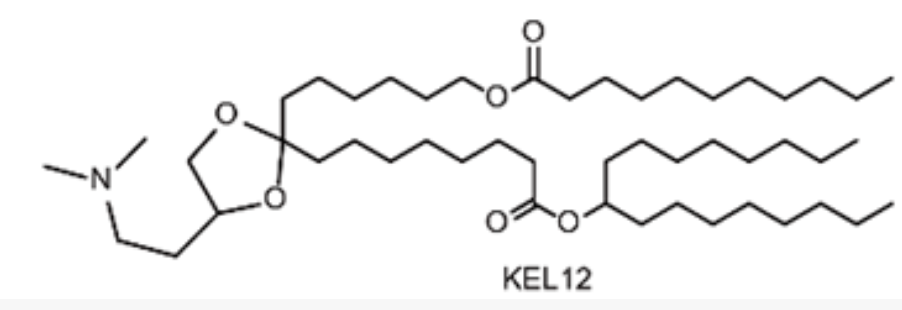
| DC67633 | Lipid KEL12 |
(4S)-KEL12 is a novel, biodegradable ionizable lipid developed for advanced mRNA vaccine delivery. It was rationally designed by incorporating both a ketal group in the linker and ester segments in the hydrophobic tails, a dual-degradable strategy aimed at enhancing its safety profile. Through iterative optimization, (4S)-KEL12 was identified as a lead candidate with an optimal pKa value of approximately 6.78, which is crucial for efficient mRNA encapsulation and endosomal release.
More description
|
|
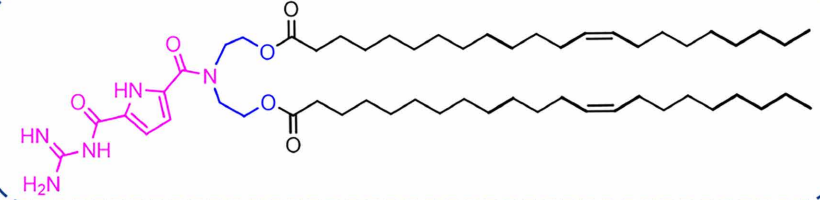
| DC67632 | Lipid GL5 |
GL5 is an ionizable guanidine-based lipid nanoparticle (G-LNP) designed for superior mRNA delivery. Its guanidinocarbonyl-pyrrole (GCP) headgroup enables pH-responsive behavior and strong mRNA binding via bidentate hydrogen bonds. The cholesterol-free GL5-3 formulation forms compact, stable nanoparticles (~90-120 nm) that exhibit excellent spleen-targeting capability after intravenous injection.GL5-LNPs efficiently deliver mRNA to antigen-presenting cells (APCs), enhancing antigen presentation and T cell activation. In cancer immunotherapy models, GL5-based mRNA vaccines provided complete tumor protection and induced durable immune memory. The platform also enables mRNA delivery to other organs like the pancreas via different administration routes, demonstrating remarkable versatility and therapeutic potential.
More description
|
|
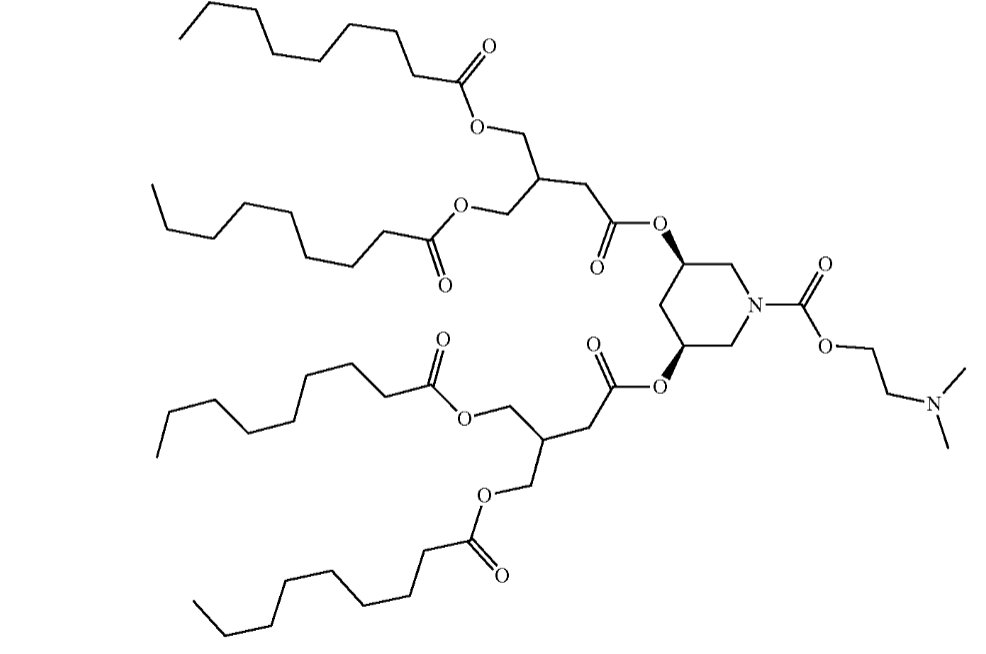
| DC67651 | CICL-238 |
Based on the data from patent US 20250127728A1, CICL-238 emerges as a highly promising ionizable lipid candidate, demonstrating notable advantages for targeted delivery applications. It achieves exceptional transfection efficiency—reaching approximately 90% of CICL-207's performance in splenic T-cells even at a reduced lipid ratio of 50% in LNP formulations. Additionally, CICL-238 exhibits minimal off-target expression in hepatocytes (<8%, comparable to CICL-207), underscoring its enhanced specificity for immune cells over liver tissues. Its optimized structure likely contributes to efficient endosomal escape and reduced Kupffer cell uptake, making it ideal for liver-related therapies (e.g., siRNA silencing for metabolic diseases) and potentially broadening applications to genetic medicine where precision and safety are paramount. Further validation in disease models could solidify its role as a versatile, low-toxicity alternative to benchmark lipids.
More description
|
|
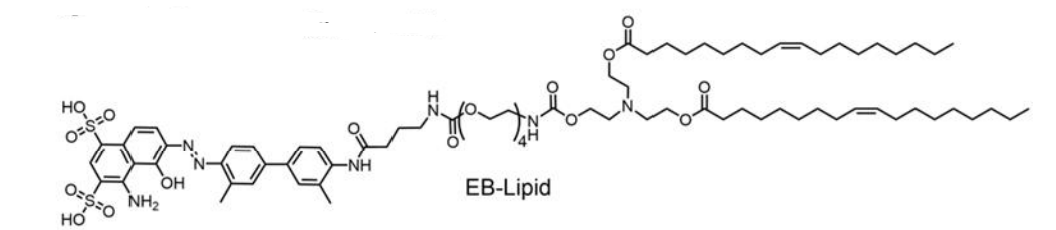
| DC67650 | EB-Lipid |
EB-Lipid is an innovatively engineered ionizable lipid designed to replace conventional PEG-lipid in mRNA vaccine formulations. Its structure comprises three key components: an Evans Blue-derived headgroup with high affinity for albumin, a tetraethylene glycol linker that enhances colloidal stability, and dual oleate tails for anchoring into lipid bilayers. This molecular design enables EB-Lipid to actively recruit endogenous albumin, forming an albumin-rich protein corona on the surface of lipid nanoparticles (LNPs). Following intramuscular administration, these albumin-bound EB-LNPs are preferentially transported through lymphatic vessels rather than entering the bloodstream, thereby avoiding hepatic accumulation and associated hepatotoxicity risks.Experimental data demonstrate that EB-LNPs achieve significantly higher accumulation in lymph nodes, where they are efficiently internalized by dendritic cells via albumin receptor-mediated endocytosis (e.g., gp60). This process enhances antigen presentation and activates robust cellular and humoral immune responses. In both tumor models (B16-OVA and HPV-associated) and infectious disease models (H1N1 and SARS-CoV-2 Omicron), EB-LNP-based mRNA vaccines elicited potent cytotoxic T-cell activation and durable neutralizing antibody production at low doses. Unlike traditional PEG-LNPs, EB-LNPs show minimal liver distribution, reduced immunogenicity, and improved safety profiles after repeated administrations.By leveraging albumin’s natural trafficking pathway, EB-Lipid represents a transformative delivery platform that combines targeted lymph node delivery with enhanced biosafety, positioning it as a promising candidate for next-generation mRNA vaccines and therapeutics.
More description
|
|
| DC67654 | ATX-012 |
ATX-012 is an ionizable cationic lipid specifically designed for mRNA delivery systems. Its unique chemical structure enables key functions in lipid nanoparticle (LNP) formulations, such as facilitating mRNA encapsulation and enhancing endosomal escape for efficient intracellular delivery.
More description
|
|
| DC67785 | KC3-OA Featured |
KC3-OA, chemically known as 3-((S)-2,2-di((Z)-octadec-9-en-1-yl)-1,3-dioxolan-4-yl)-N,N-dimethylpropan-1-amine, is an ionizable cationic lipid (ICL) optimized for lipid nanoparticle (LNP) formulations in nucleic acid delivery, particularly for mRNA vaccines. It features a unique structure with mono-unsaturated alkyl chains (C18:1), which enhances oxidative stability compared to polyunsaturated analogs like KC3, while maintaining efficient membrane fusion and endosomal escape capabilities. In LNP compositions, KC3-OA is typically incorporated at 46–54 mol% of total lipids, with an N/P ratio of 4–6 relative to mRNA, ensuring high encapsulation efficiency and transfection potency.
Experimental data demonstrate that KC3-OA-based LNPs achieve superior mRNA expression in human dendritic cells, outperforming alternatives like KC3-PA or KC3-01 in both in vitro and in vivo models. For instance, in FIG. 2, KC3-OA LNPs showed ~2-fold higher mCherry expression at low mRNA doses (0.1 μg/mL) due to improved cellular uptake and reduced degradation. Its synergy with anionic phospholipids like DPPS (5 mol%) further enhances dendritic cell targeting via receptor-mediated internalization, leading to robust CD4+ and CD8+ T-cell responses against Mycobacterium tuberculosis antigens. This balance of stability, efficiency, and immunogenicity makes KC3-OA a leading candidate for next-generation vaccines.
More description
|
|
| DC67984 | L31(Lipid 31) Featured |
L31 is identified as a novel, proprietary ionizable cationic lipid that serves as the critical functional component within lipid nanoparticles (LNPs) engineered for CRISPR/Cas9 genome editing in head and neck squamous cell carcinoma (HNSCC). It was selected from a screened library of lipids for its superior performance. LNPs formulated with L31 exhibited excellent physicochemical properties, including a uniform size of 80-100 nm, low polydispersity, and high encapsulation efficiency (>85%) for both Cas9 mRNA and sgRNA. In vitro, L31-based LNPs demonstrated outstanding therapeutic efficacy, achieving approximately 68% gene editing of the oncogene SOX2 and an 88% reduction in cancer cell viability.For in vivo applications, L31-LNPs were further functionalized with anti-EGFR antibodies using the ASSET linker strategy to create targeted nanoparticles (tLNPs). This modification enhanced specific uptake by tumor cells. In a xenograft mouse model, intratumoral injection of these targeted L31-cLNPs co-encapsulating Cas9 mRNA and sgSOX2 led to potent tumor growth inhibition (90%) and a significant increase in survival, with tumor disappearance observed in half of the treated mice. In conclusion, L31 is a highly efficient ionizable lipid that forms the foundation of a potent targeted LNP platform for precise CRISPR-based cancer therapy against solid tumors.
More description
|
.png)
|
| DC67981 | Diamino lipid DAL4 Featured |
Diamino lipid DAL4 is diamino lipid for the preparation of lipid nanoparticles (LNPs) encapsulated with mRNAs encoding cytokines including IL-12, IL-27 and GM-CSF. Diamino lipid DAL4 delivers mRNA to tumor cells to exert anti-tumor activity.
More description
|
|
| DC67988 | KT-001 Featured |
KT-001 is a novel ionizable cationic lipid disclosed in patent US 2026/0007612 A1
More description
|
|
| DC67983 | XH-07 Featured |
XH-07 is an innovative ionizable cationic lipid that forms the backbone of the JCXH-211 lipid nanoparticle (LNP) delivery system. This complex is engineered to encapsulate and deliver self-replicating RNA (srRNA) encoding interleukin-12 (IL-12), a potent immunostimulatory cytokine. The LNP formulation featuring XH-07 exhibits optimal physicochemical properties, such as a mean particle size of approximately 82.12 nm with low polydispersity, and a near-neutral zeta potential around -3.181 mV, which facilitates stable circulation and efficient cellular uptake upon intravenous administration. Upon delivery, the srRNA leverages the host cell's machinery to produce sustained levels of IL-12p70, as demonstrated in B16F10 tumor-bearing mice, where a single dose led to peak cytokine production in sera and tumors. This induced IL-12 expression activates T cells and NK cells, generating a robust antitumor response. In murine models of melanoma and breast cancer, JCXH-211 monotherapy resulted in significant tumor regression and complete responses in some subjects, and it synergized with anti-PD-1 therapy to enhance efficacy. Importantly, the safety profile was acceptable, with transient liver enzyme elevations in mice that normalized quickly, and no significant adverse events in cynomolgus monkeys after repeated dosing, as evidenced by stable clinical observations and pathology tests. Thus, XH-07 is pivotal for enabling the safe and effective delivery of IL-12 encoding RNA, positioning JCXH-211 as a promising cancer immunotherapy.
More description
|
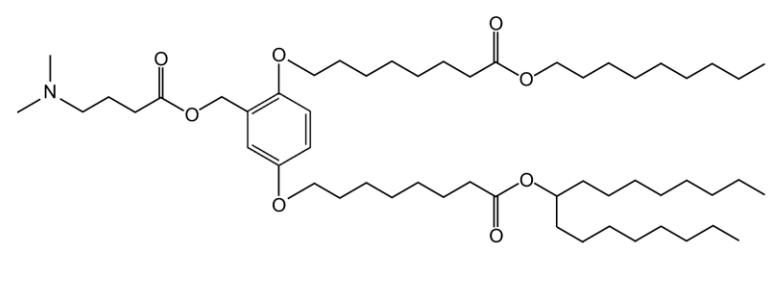
|
| DC67662 | Lipid 48 Featured |
Lipid 48 is a leading ionizable lipid designed for therapeutic nucleic acid delivery. Its key function is to form the core of lipid nanoparticles (LNPs) that efficiently encapsulate and deliver cargoes like mRNA and CRISPR guide RNAs into cells. Its optimized structure allows it to remain neutral in the bloodstream for low toxicity but become positively charged in acidic cellular compartments (endosomes), where it disrupts the membrane to release the therapeutic payload. Data from the patent demonstrates its superior profile: it achieves high gene editing efficiency (e.g., ~80% indel rates in vitro and 16.2% in vivo in mouse liver) while maintaining low cytotoxicity (cell viability >80% at effective doses), establishing it as an ideal candidate for gene therapy applications due to its exceptional balance of potency and safety.
More description
|
.png)
|
| DC67994 | 244-9-cis Featured |
244-9-cis is a novel ionizable lipid disclosed in United States Patent US 2026/0014075 A1, specifically engineered for advanced lipid nanoparticle (LNP) delivery systems. Its distinctive molecular architecture features biodegradable ester bonds, which contribute to excellent physicochemical properties such as a near-neutral surface charge (approximately -3 mV) for improved biocompatibility, an optimal pKa of about 6.2 to facilitate endosomal escape, and consistently high nucleic acid encapsulation efficiency exceeding 90%. In vivo studies confirm significantly enhanced delivery to hepatocytes and markedly higher therapeutic protein expression compared to control formulations, positioning 244-9-cis as a promising candidate for next-generation genetic medicines.
More description
|
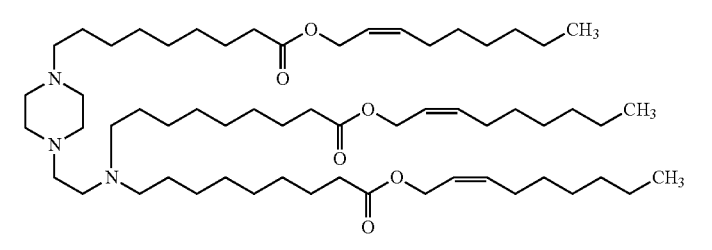
|
| DC67995 | Lipid 22 Featured |
Compound 22, as detailed in United States Patent US 2026/0014089 A1, is a bifunctional ionizable lipid engineered for precision drug delivery. Its structure integrates a monosaccharide targeting headgroup, designed to bind specifically to DC-SIGN receptors on dendritic cells, via a sophisticated linker connected to a biodegradable lipid anchor. This design enables it to serve as a key component of lipid nanoparticles (LNPs), forming a targeted delivery system. By leveraging the specific carbohydrate-receptor interaction, these LNPs are preferentially internalized by dendritic cells, critical for initiating adaptive immune responses. In vivo studies from the patent, such as the biodistribution data shown in Figure 5, confirm effective accumulation in lymphoid tissues like the spleen and lymph nodes. Consequently, this targeted delivery enhances the potency of encapsulated payloads (e.g., mRNA vaccines) by ensuring professional antigen presentation, eliciting a stronger and more specific immune response—evidenced by higher neutralizing antibody titers—making it a powerful tool for next-generation vaccines and therapeutics.
More description
|
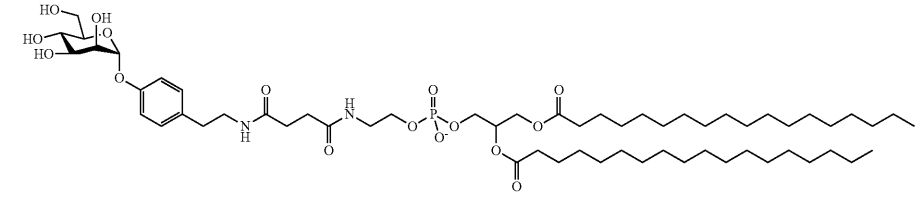
|
| DC60922 | Lipid AP60 Featured |
AP60 is a novel, biomimetic ionizable lipid, identified as the lead compound from a library of 67 aminophosphonate-derived lipids. Inspired by the structure of natural phospholipids, it forms the core component of lipid nanoparticles within the CROSS delivery platform. AP60-based LNPs demonstrate superior efficiency in delivering mRNA and circular RNA to neuronal cells and astrocytes compared to the clinically used MC3 LNP. In a mouse model of spinal cord injury, AP60 LNPs achieved significantly higher protein expression at the lesion site (13.7-fold locally, 4.6-fold intravenously) while concurrently reducing off-target accumulation in the liver by nearly 5-fold. This indicates improved targeting to the central nervous system injury site and a potentially better safety profile. Its cellular uptake is primarily mediated by clathrin-mediated endocytosis and macropinocytosis, followed by efficient endosomal escape. By encapsulating therapeutic circular RNAs encoding Sox2, Ascl1, and GDNF, AP60 LNPs enable effective in vivo reprogramming and neuroprotection, leading to significant functional recovery of bladder and locomotor functions after spinal cord injury.
More description
|
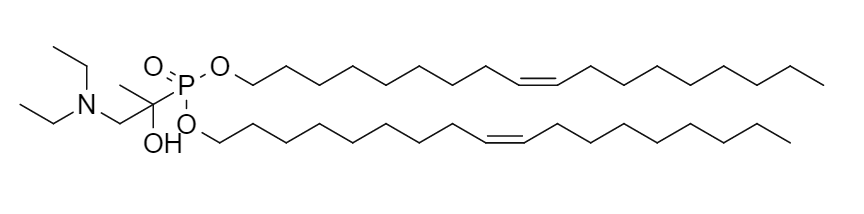
|
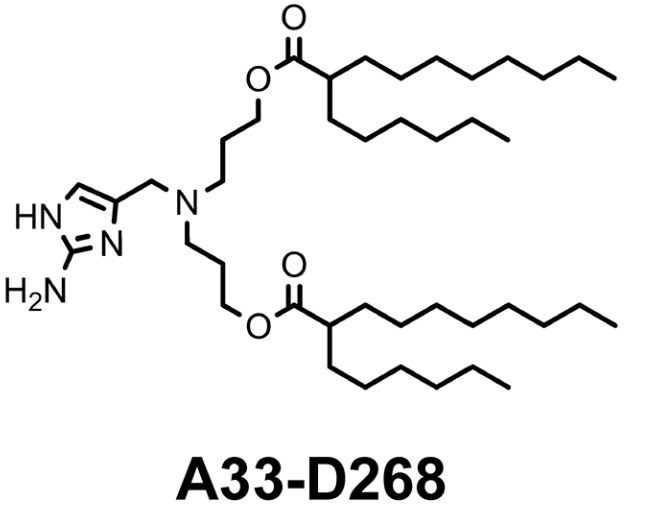
| DC60872 | Lipid A33-D268 |
A33-D268 is an ionizable lipid derived from a 2-aminoimidazole (AM) core, featuring an asymmetric hydrophobic tail structure designed to optimize mRNA interactions. Selected as the top candidate from a 21-lipid library through molecular dynamics (MD) simulations, it demonstrated superior performance: low root mean square deviation (RMSD) and the smallest radius of gyration (Rg), indicating tight mRNA compaction, alongside moderate electrostatic energy and above-average hydrogen bonding for stable encapsulation. Formulated into LNPs, it achieves efficient muscle-specific transfection post-intramuscular injection, rivaling commercial ALC-0315 LNPs. Crucially, it exhibits 29-fold lower hepatic off-target expression, attributed to its inability to leverage serum protein coronas for liver tropism.
More description
|
|
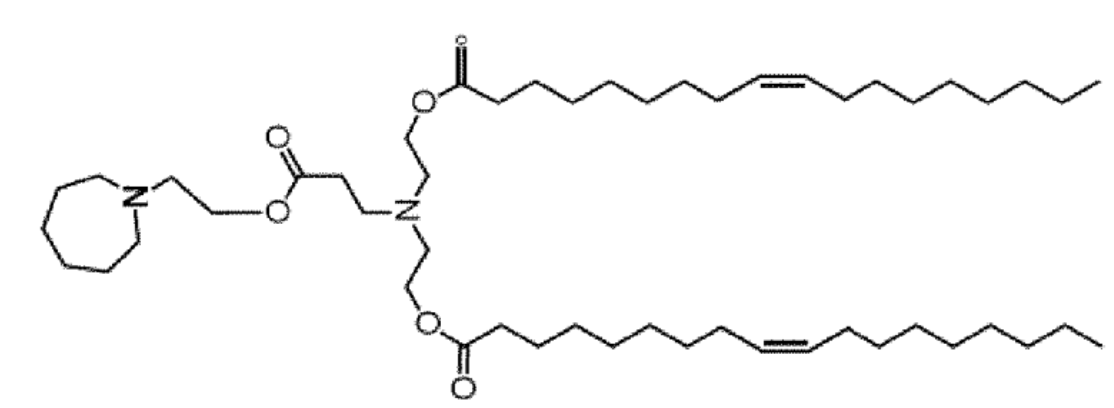
| DC68020 | Lipid OC7 Featured |
Lipid OC7, as described in the patent WO2022207938A1, is a novel ionizable lipid that serves as the core functional component of the saNppa-LNP delivery system. Its key innovation lies in its unique biodegradable structure featuring an internal ester bond. Under typical physiological conditions, this bond hydrolyzes, triggering a charge shift from a cationic form that complexes nucleic acids to a zwitterionic form that releases them. This property is central to its role in enabling long-acting self-amplifying RNA (saRNA) therapies. Specifically, OC7 facilitates immune stealth by mitigating early interferon responses, supports sustained and efficient intracellular replication of saRNA even at low doses, and enables therapeutic protein expression that persists for over 28 days from a single administration. This combination of efficient delivery, controlled release, and extended duration of action makes OC7-based LNPs a promising platform for long-term treatments, such as for myocardial infarction, as demonstrated in the referenced research.
More description
|
|
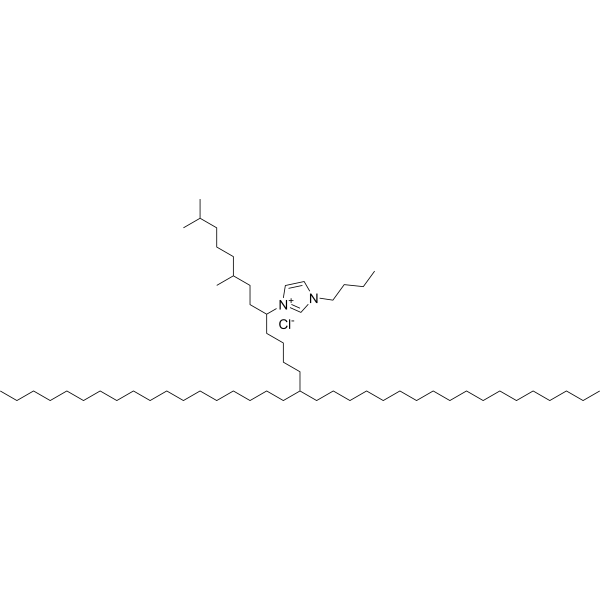
| DC67134 | IM21.7c |
|
|
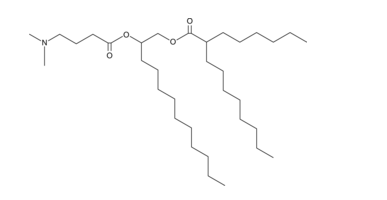
| DC66221 | E12CA1A3 |
E12CA1A3 is an ionizable cationic lipid (pKa = 6.4) that has been used in the generation of lipid nanoparticles (LNPs) for the delivery of mRNA in vitro and in vivo.1 LNPs containing E12CA1A3 and encapsulating an mRNA reporter induce luciferase reporter expression in mouse bone marrow-derived dendritic cells (BMDCs) and mice. LNPs containing E12CA1A3 are cleared more rapidly from the liver than LNPs containing DLin-MC3-DMA (Item No. 34364) in mice.
More description
|
|
| DC67283 | C14-490 |
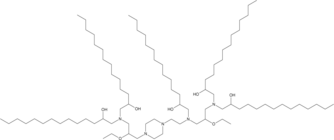
C14-490 is an ionizable cationic lipid (pKa = 5.94).1 It has been used in the generation of lipid nanoparticles (LNPs) for the delivery of mRNA in vivo.C14-490-containing LNPs accumulate primarily in the fetal mouse liver and, to a lesser extent, in the lungs, intestine, and brain after vitelline vein injection on gestational day 16 (E16).1 CD45-functionalized LNPs containing C14-490 and encapsulating mRNA encoding GFP transfect Jurkat cells, which constitutively express the CD45 receptor (CD45R).C14-490-containing CD45-functionalized LNPs encapsulating Cre mRNA mediate genome modulation in bone marrow hematopoietic stem cells (HSCs) for at least four months after in utero injection in R26mT/mG mice at E13.5.
More description
|
|
| DC67530 | HY-501 Featured |
HY-501 is a next-generation cationically ionizable lipid engineered for high-efficiency RNA delivery developed by Biontech. Formulated at 40–50 mol% in lipid nanoparticles (LNPs) alongside DSPC, cholesterol, and polysarcosine-conjugated lipid C14pSar23, HY-501 yields uniform, stable particles (80–100 nm) with >90% RNA encapsulation. It demonstrates superior in vivo performance: driving 2-fold higher protein expression than benchmark lipids (EA-405/HY-405) in muscle tissue, minimizing off-target liver accumulation, and reducing immunogenic risks (near-zero complement activation and <5% hemolysis). Preclinically, HY-501-based LNPs encoding SARS-CoV-2 spike protein elicit potent neutralizing antibodies and T-cell responses, underscoring its utility in precision vaccines. Its combination of scalable synthesis, exceptional transfection efficiency, and biosafety establishes HY-501 as a transformative vector for therapeutic RNA delivery.
More description
|

|
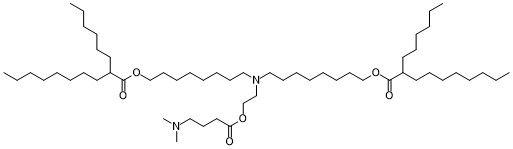
| DC82125 | lipid 14 Featured |
LIPID 14 is a novel ionizable lipid used for mRNA delivery.In 2021, Elia et al. used lipid 2 LNPs and lipid 14 LNPs to deliver mRNA encoding SARSCoV-2 human Fc-conjugated receptor binding domain (RBDhFc
mRNA). While both lipid 274 LNP RBD-hFc mRNA and
lipid 14 LNP RBD-hFc mRNA induced equal cellular and
humoral responses in mice at an mRNA dose of 5 μg, only lipid
14 LNP RBD-hFc mRNA exhibited strong immunogenicity
following intradermal administration. Both intradermal administration
and intramuscular administration of lipid 14 LNPs
could activate antigen presenting cells (APCs), thus inducing
cellular responses.
More description
|
|
| DC67549 | ORNA lipid 144(1-C) |
ORNA lipid 144 is a novel ionizable lipid engineered for splenic RNA delivery developed by ORNA Therapeutics, featuring a biodegradable structure with a protonatable tertiary amine headgroup and ester-linked branched C14 alkyl chains. This design enables exceptional spleen-targeting capability, demonstrated by 3-fold higher luciferase expression in the spleen compared to benchmark lipids and near-complete B-cell depletion when delivering anti-CD19 CAR circRNA. It forms highly stable lipid nanoparticles maintaining homogeneous size (60–80 nm) and low polydispersity across diverse manufacturing conditions and buffer systems. Rapid clearance from the liver and spleen minimizes off-target accumulation, while high circRNA encapsulation efficiency (>90%) and pH-dependent endosomal escape make it ideal for immunotherapies and vaccines requiring precise splenic bioavailability and sustained efficacy.
More description
|

|
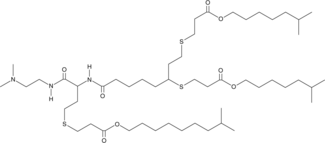
| DC67460 | CP-LC-1422 |
Derived from the natural amino acid homocysteine, CP-LC-1422 is an ionizable cationic lipid that enables robust in vivo delivery of various RNA forms (mRNA, cRNA, and saRNA), driving high protein expression. When formulated into LNPs (50/38.5/10/1.5 molar ratio of ionizable lipid/cholesterol/DOPE/PEG-lipid), it achieves superior spleen-specific targeting compared to commercial options through intravenous administration, while maintaining an excellent safety profile.
More description
|
|
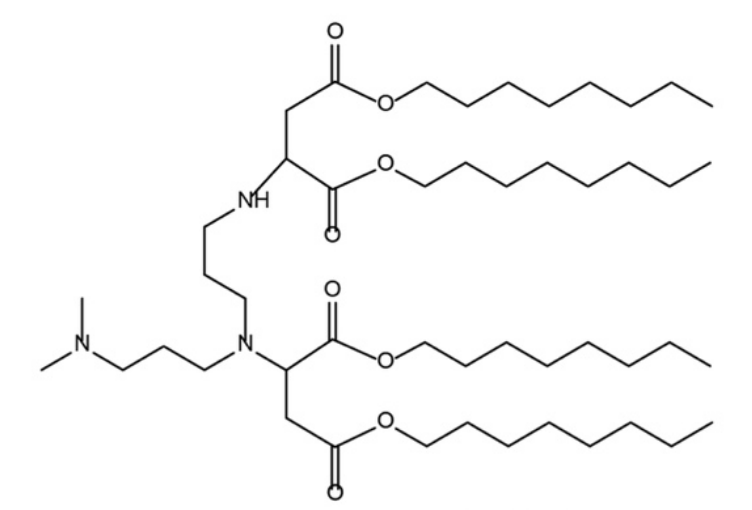
| DC67298 | Lipid 5D8 Featured |
Lipid 5D8 is a novel biodegradable ionizable lipid (IL) developed through a combinatorial chemistry strategy to overcome the limitations of conventional lipid nanoparticles (LNPs) in mRNA delivery. Synthesized via a one-step, solvent-free Michael addition reaction between amine and thiol monomers, 5D8 features asymmetric lipid tails and a biodegradable ester backbone, ensuring both structural versatility and reduced toxicity. In preclinical studies, 5D8-based LNPs demonstrated exceptional liver-targeting efficiency and mRNA delivery performance. A single intravenous dose (1 mg/kg) achieved 61% CRISPR-Cas9-mediated editing of the TTR gene in mice, reducing serum TTR protein by 90%, outperforming benchmark lipids like C12-200 (51% editing). Moreover, 5D8 enabled efficient delivery of base editors (ABE8.8 and CBE4max), achieving 42% PCSK9 editing (74% serum protein reduction) and correcting hereditary tyrosinemia in mice, significantly extending survival. Beyond gene editing, 5D8 LNPs effectively delivered siRNA (complete serum TTR clearance at 0.05 mg/kg) and enhanced hepatocyte targeting by enriching apolipoprotein E on particle surfaces. Crucially, 5D8 exhibited superior biocompatibility with no hepatotoxicity (normal ALT/AST levels), contrasting traditional LNPs. Its rapid biodegradability and "plug-and-play" design make 5D8 a versatile platform for mRNA therapeutics, holding broad potential for treating genetic disorders, cardiovascular diseases, and beyond. This innovation represents a critical advancement toward safer, high-efficiency clinical translation of gene-editing therapies.L
More description
|
|
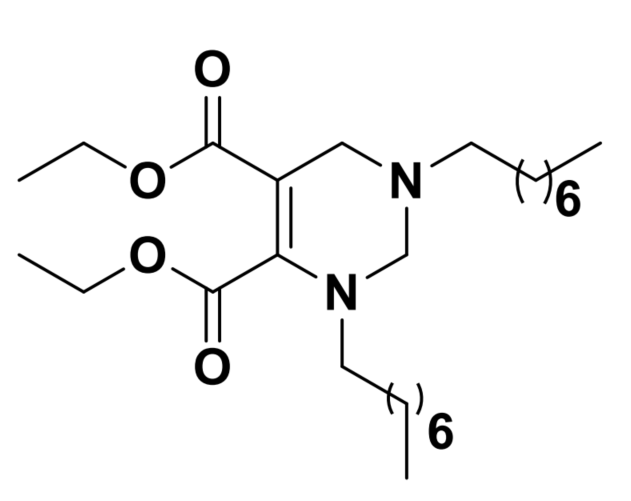
| DC60671 | THP1 |
THP1 is a tetrahydropyrimidine ionizable lipid for mRNA delivery. THP1 demonstrates higher transfection efficiency comparable to DLin-MC3-DMA (MC3). THP1 LNPs also demonstrates the ability to edit genes in specific liver tissues in a tdTomato transgenic mouse model.
More description
|
|
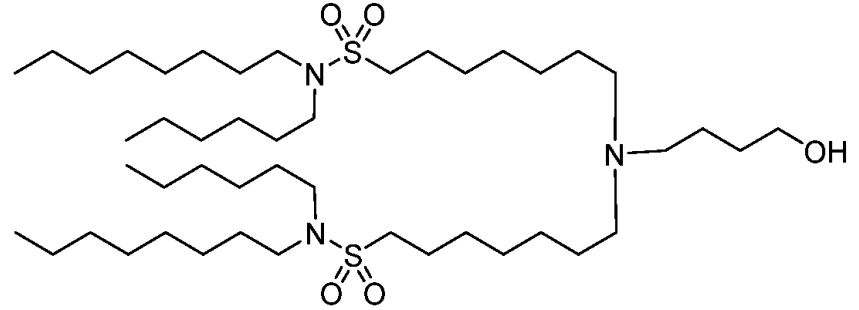
| DC67281 | BNT-51 Featured |
BNT-51 is an ionizable thiolipid developed by Biontech, characterized by its sulfur-containing moieties and a multiarm dendron-like architecture. Synthesized via reactions between amine-containing compounds and sulfur-based halides or sulfonates, it forms stable lipid nanoparticles (LNPs) optimized for mRNA delivery. The LNPs exhibit uniform particle size (80–100 nm, PDI <0.2), near-neutral zeta potential, and high mRNA encapsulation efficiency (>90%), while maintaining payload integrity through freeze-thaw cycles and extended storage. In vitro, BNT-51 demonstrates low cytotoxicity (>80% cell viability in C2C12, HepG2, and HEK293 cells) and superior transfection efficiency compared to conventional lipids, particularly in immune cells such as CD4+/CD8+ T cells within PBMCs. Its modular design allows integration of stealth lipids (e.g., PEG or vitamin E derivatives) to prolong circulation time and minimize immune activation, as evidenced by low hemolysis and complement activation risks. In vivo, BNT-51-based LNPs enable targeted mRNA delivery to splenic macrophages, achieving potent genome editing (e.g., Cre mRNA) and therapeutic protein expression (e.g., BACH1) in preclinical models. With its tunable structure, robust stability, and cell-specific tropism, BNT-51 holds promise for advancing mRNA therapeutics in gene editing, cancer immunotherapy, and regenerative medicine, offering a versatile platform for next-generation nanomedicine.
More description
|
|
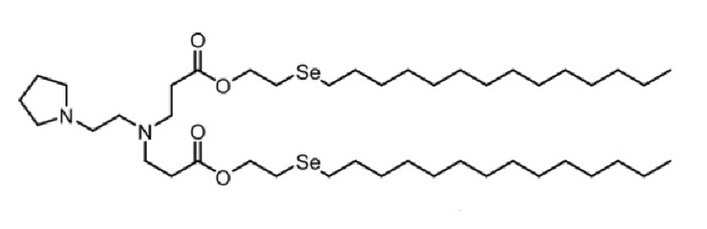
| DC60494 | 76-O17Se |
76-O17Se is a lipidoid for the efficient delivery of antiCD19 mRNA CAR to murine primary macrophages. 76-O17Se is more efficient than delivery with lipofectamine 2000 (LPF2K) or MC3
More description
|
|
| DC67557 | Tidal Lipid 40 |
Tidal Lipid 40is an ionizable cationic lipid engineered to deliver RNA with high precision to immune cells like macrophages. Based on Tidal Therapeutics' patent US 20250205169A1, Its pH-responsive design shifts from a +8 mV charge at pH 5.5 (enabling endosomal escape) to near-neutral at pH 7.4 (reducing off -target binding), ensuring efficient intracellular release while maintaining blood stability. In lipid nanoparticles, Lipid 40 achieves 65% transfection efficiency in human macrophages—surpassing benchmarks like ALC-0315—and protects >95% of RNA payloads from degradation. Critically, it maintains particle integrity after freeze-thaw cycles with minimal size drift (<5 nm) and excels in in vivo targeting, driving potent gene expression in tumor-associated macrophages while avoiding liver/spleen accumulation. This combination of precision delivery, stability, and low toxicity makes it ideal for immunotherapies, such as reprogramming M2 macrophages to anti-tumor M1 states.
More description
|

|
| DC67544 | HCQ Lipid 4(HCQ-4) |
HCQ-4 is a rationally engineered ionizable lipid derived from hydroxychloroquine (HCQ), featuring a ditetradecylamine-derived twin-C14 saturated hydrocarbon tail linked to the HCQ headgroup via a succinic acid spacer. Synthesized through a three-step route involving HCQ deprotonation, ditetradecylamine carboxylation, and EDC/DMAP-mediated amidation, this lipid forms the core of optimized lipid nanoparticles (LNPs) at a molar ratio of 60:10:40:0.5 (HCQ-4:DOPE:cholesterol:DMG PEG2000). The structure enables dual functionality: (1) Spleen-selective mRNA delivery (2.3-fold higher splenic vs. hepatic transfection) via 80-100 nm particle size, near-neutral charge (-3 mV), and low PEG density, facilitating immune cell uptake; (2) Tumor microenvironment modulation through HCQ-mediated repolarization of M2 macrophages to antitumor M1 phenotype (iNOS+ cells ↑2.5-fold, CD206+ cells ↓60%). This bifunctional design synergistically enhances mRNA cancer vaccine efficacy, demonstrating superior prophylactic/therapeutic antitumor activity and antimetastatic effects compared to clinical benchmarks like MC-3 LNP.
More description
|

|
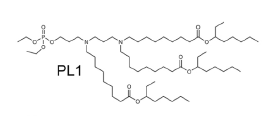
| DC80068 | LIPID PL1 |
PL1 is a novel biomimetic phospholipid. PL1 nanoparticle delivery of costimulatory
receptor CD137 mRNA improved the immunotherapy with an
anti-CD137 Ab to some extent in both tumor models with better
results obtained in the B16F10 melanoma model as compared to
the A20 lymphoma model.
More description
|
|