 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
| Cat. No. | Product Name | Field of Application | Chemical Structure |
|---|---|---|---|
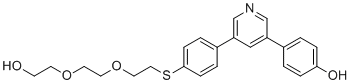
| DC74440 | CW85319 Featured |
CW85319 is a small-molecule compound that enhances Axin2's interaction with GSK3β, shows a high-affinity binding with Axin2 with Kd of 5.8 uM.
More description
|
|
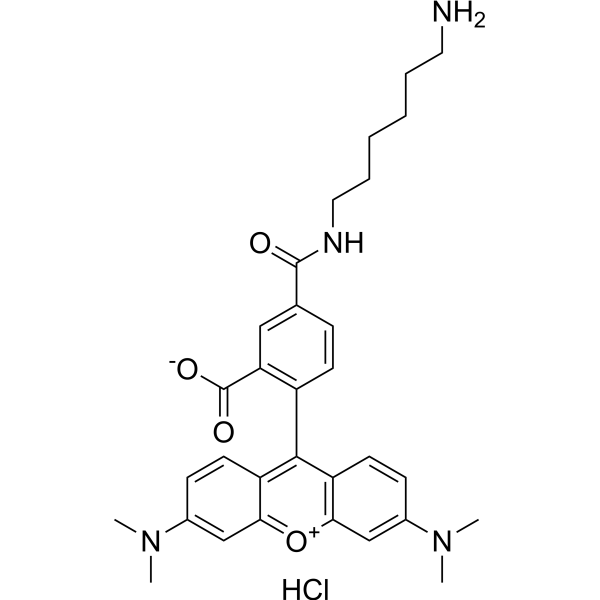
| DC68117 | TAMRA amine, 5-isomer Featured |
TAMRA amine, 5-isomer hydrochloride is a rhodamine dye with excitation/emission maximum 553/575 nm. The amine group is reactive with carboxylic acids, activated NHS esters, carbonyls (ketone, aldehyde) etc.
More description
|
|
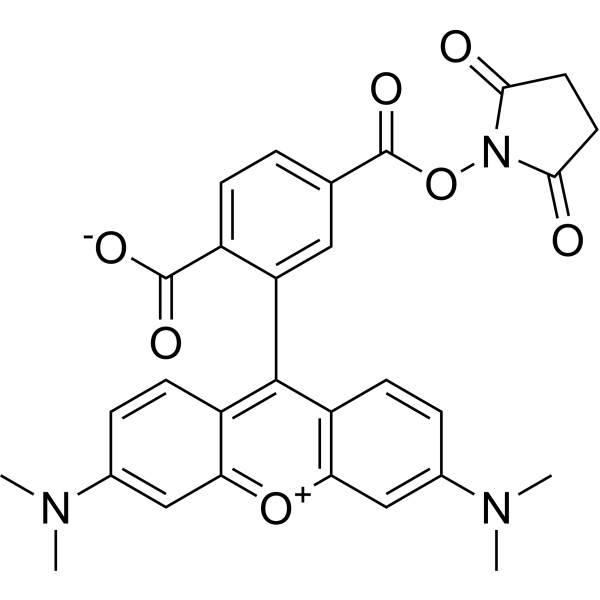
| DC68116 | 6-TAMRA-SE Featured |
6-TAMRA-SE (6-TAMRA-NHS ester) is a fluorescent dye carrying the amine reactive group. 6-TAMRA-SE is one of the traditional fluorophores used for automated DNA sequencing.
More description
|
|
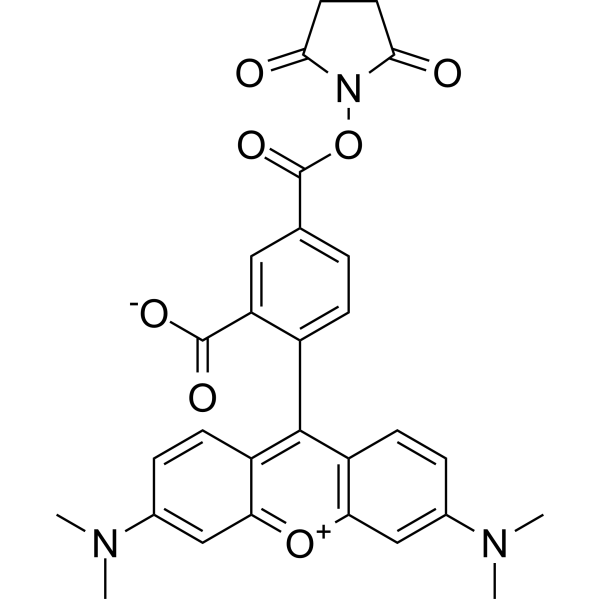
| DC67950 | 5-TAMRA NHS Featured |
5-TAMRA NHS is an amine-reactive fluorescent agent, and its conjugate produces bright, pH-insensitive orange-red fluorescence with good photostability (Ex/Em = 565/580 nm).
More description
|
|
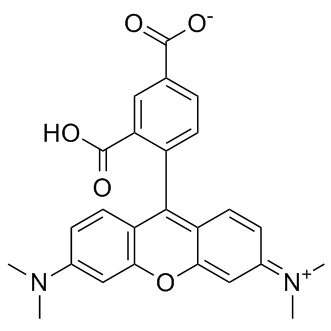
| DC9373 | 5-TAMRA Featured |
The amine-reactive 5-TAMRA, SE and its conjugates yield bright, pH-insensitive orange-red fluorescence (approximate excitation/emission maxima ~546/579) with good photostability.
More description
|
|
| DC9374 | 6-TAMRA Featured |
Tetramethylrhodamine (TMR, TRITC) has been a widely used fluorophore for preparing bioconjugates, especially fluorescent antibody and avidin derivatives used in immunochemistry.
More description
|

|
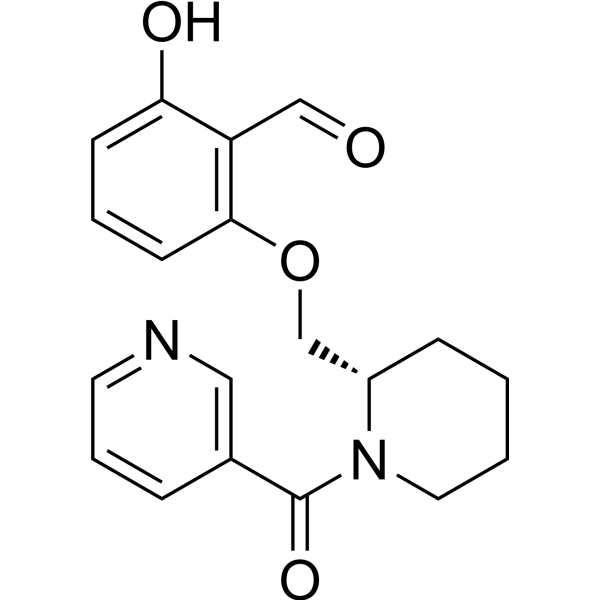
| DC72883 | GBT1118 Featured |
GBT1118 is a potent and orally active allosteric modifier of hemoglobin oxygen affinity. GBT1118 increases tolerance to severe hypoxia.
More description
|
|
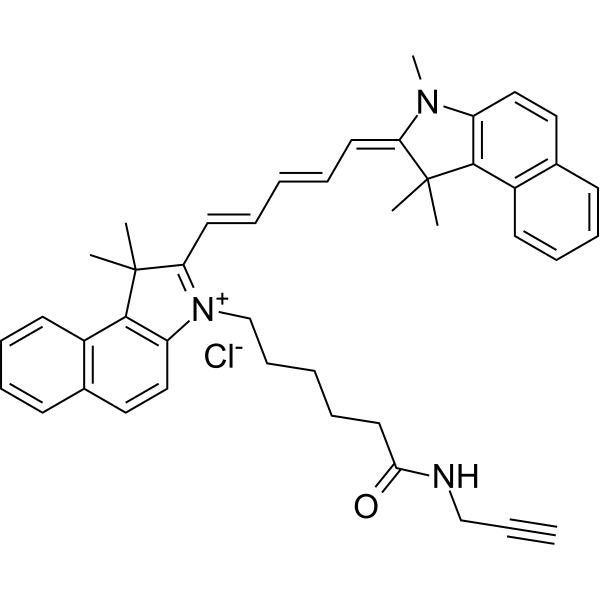
| DC33878 | Cyanine5.5 alkyne Featured |
Far red / near infrared dye alkyne for Click Chemistry labeling. Cyanine5.5 is an analog of Cy5.5, a popular fluorophore which has been widely used for various applications including intact organism imaging. This reagent can be conjugated with azido groups under mild copper catalyzed Click Chemistry conditions.
This reagent is soluble in organic solvents, but mixtures of water with small percent of DMSO can be used for efficient conjugation. Cyanine5.5 alkyne can also be used for the labeling of small molecules with this far red/NIR dye.
More description
|
|
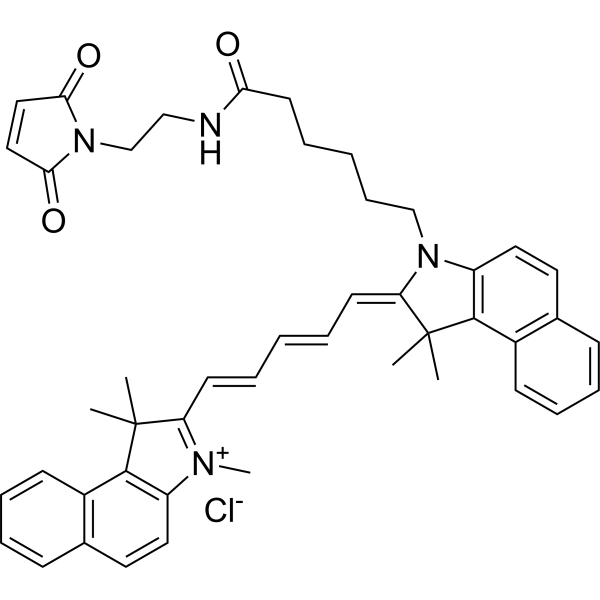
| DC33898 | Cyanine 5.5 maleimide Featured |
Cyanine5.5 maleimide is a thiol reactive dye which is capable of selective labeling of sulfhydryl groups in proteins, an analog of Cy5.5? maleimide.
Near infrared emission of Cyanine5.5 makes this dye suitable for bioimaging applications.
Cyanine5.5 can replace Cy5.5?, Alexa Fluor 680, and DyLight 680.
More description
|
|
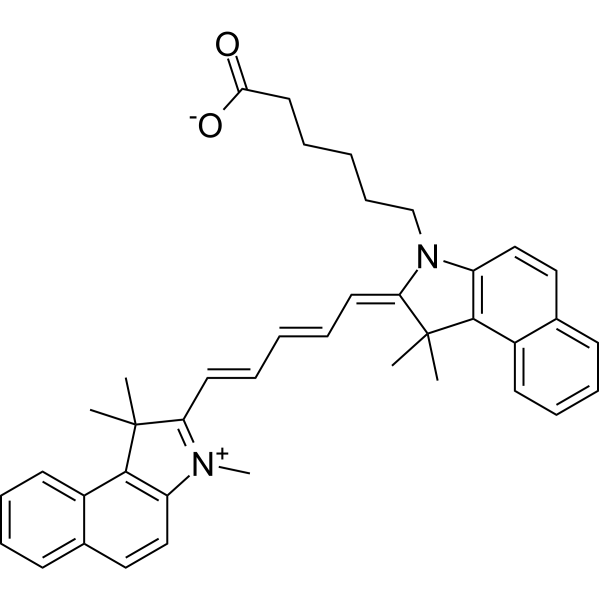
| DC33638 | Cy5.5 carboxylic acid Featured |
CY5.5 carboxylic acid, also known as Cyanine5.5 carboxylic acid, is a Far-red emitting fluorescent dye with Ex/Em wavelength 673nm/707nm. CY5.5 carboxylic acid can be considered non-reactive for most applications and used as a control or reference sample, and for instrument calibration.
More description
|
|
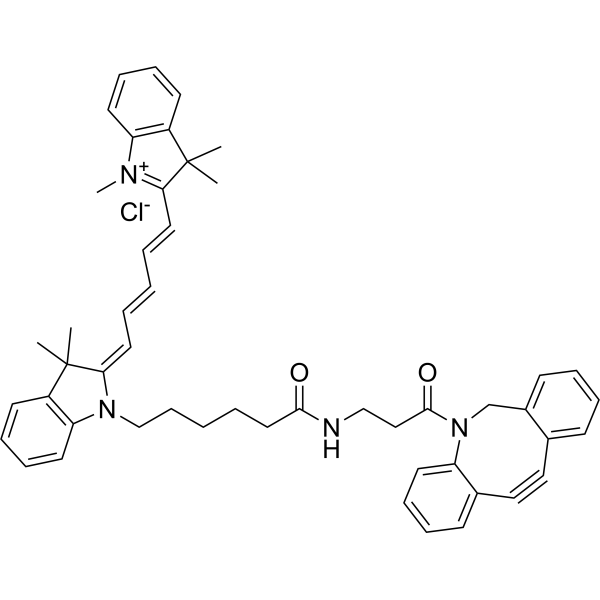
| DC68115 | Cy5 DBCO Featured |
Cy5 DBCO chloride is an azide reaction probe and the addition of DBCO molecules allows the imaging of azide-labelled biomolecules by a copper-free "Click Chemistry" reaction[1]. Cy5 DBCO (chloride) is a click chemistry reagent, it contains a DBCO group that can undergo strain-promoted alkyne-azide cycloaddition (SPAAC) with molecules containing Azide groups.
More description
|
|
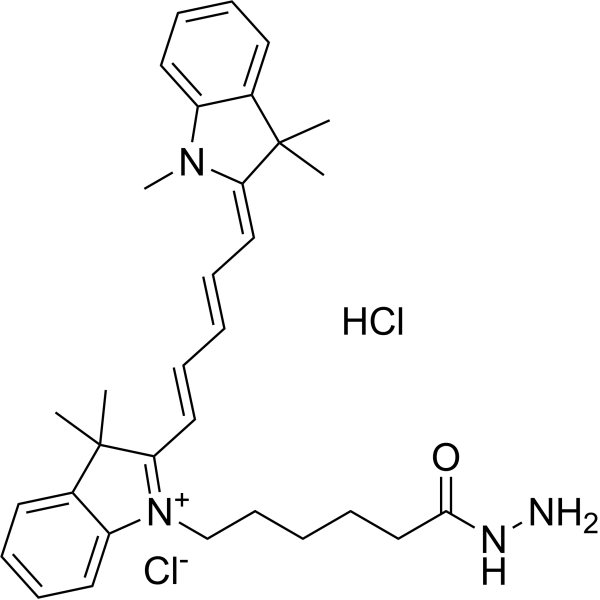
| DC68114 | Cyanine5 hydrazide Featured |
Cy 5 hydrazide (Cyanine5 hydrazide) is a fluorescent dye for aldehydes and ketones. Cy 5 hydrazide can be used for labeling proteins.
More description
|
|
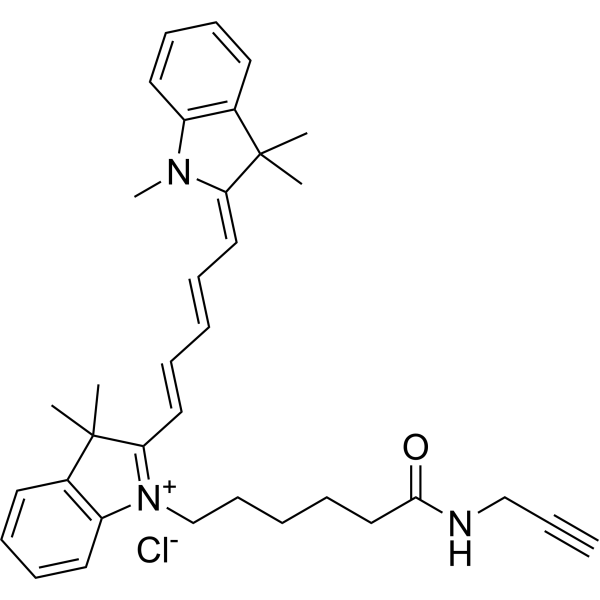
| DC33877 | Cyanine5 alkyne Featured |
Cyanine5 alkyne for Click Chemistry, an analog of Cy5 alkyne. With this product, deeply colored, and photostable Cyanine5 fluorophore can be attached to various molecules via Click Chemistry reaction with azides.
This alkyne is non-water soluble, but it can be dissolved in DMF or DMSO prior to reaction, and added to aqueous reaction mixture. With our labeling protocol, the labeling reaction is very efficient, and high-yielding.
Various substrates bearing azides can be used for the labeling, including azido-labeled biomolecules, polymers, and solid surfaces.
More description
|
|
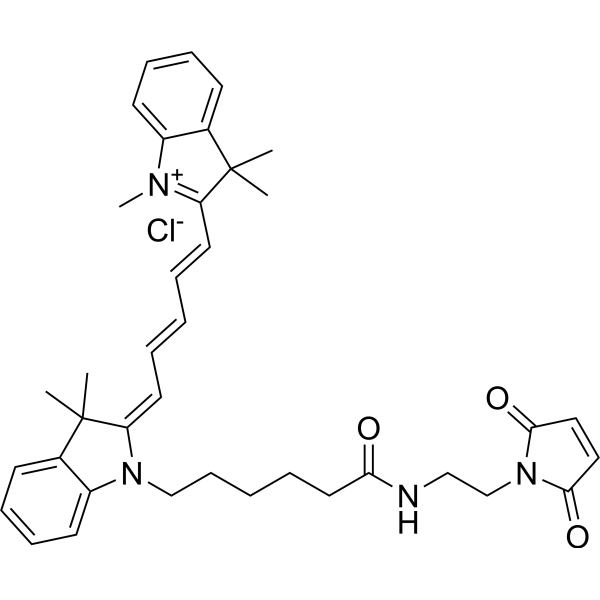
| DC33897 | Cyanine5 maleimide Featured |
Cyanine5 maleimide is a mono-reactive dye which selectively couples with thiol groups (for example, with cysteines in peptides and proteins) to give labeled conjugates.
Cyanine5 is an analog of Cy5?, a common fluorophore which is compatible with various instrumentation like microscopes, imagers, and fluorescence readers.
For the labeling of antibodies and sensitive proteins we recommend to use the water soluble sulfo-Cyanine5 maleimide.
More description
|
|
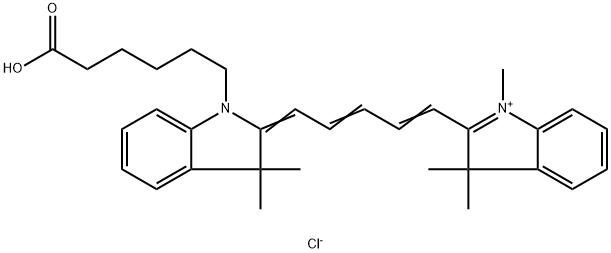
| DC33932 | Cyanine5 acid Featured |
Non-activated carboxylic acid, an analog of Cy5? free carboxylic acid. Contains Cyanine5 fluorophore.
This dye has limited water solubility, but can be dissolved in mixtures of water with organic phase (DMF, DMSO, alcohols) to obtain useful concentrations of the material in solution. Water-soluble version is available.
This molecule can be considered non-reactive dye for the use in control samples, and for instrument calibration. For coupling with amines and protein labeling, consider using Cyanine5 NHS ester, or water-soluble sulfo-Cyanine5 NHS ester.
More description
|
|
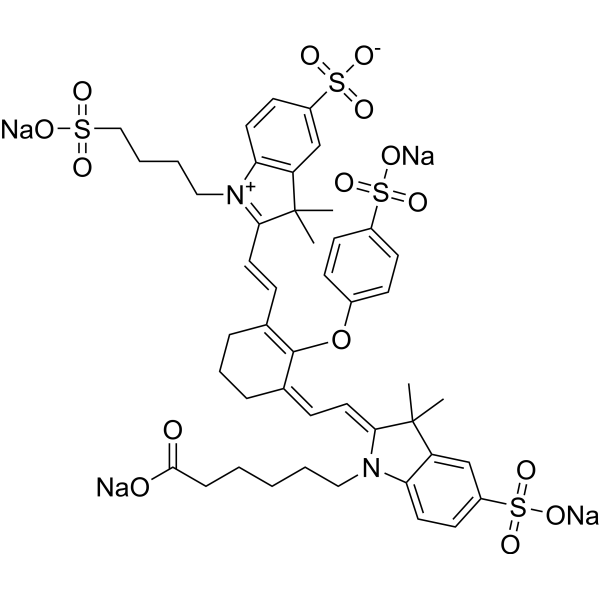
| DC68113 | 800CW acid (IRDye 800CW acid equivalent) Featured |
800CW acid is an orally active near-infrared (NIR) dye (excitation/emission 774/789 nm). 800CW acid can be used for intestinal permeability testing. 800CW acid can be used for in vivo imaging studies.
More description
|
|
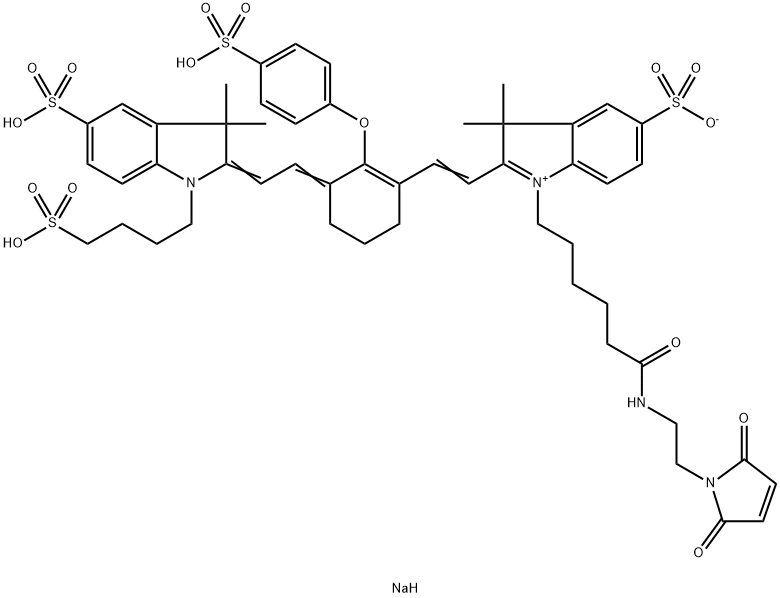
| DC68112 | 800CW maleimide Featured |
|
|
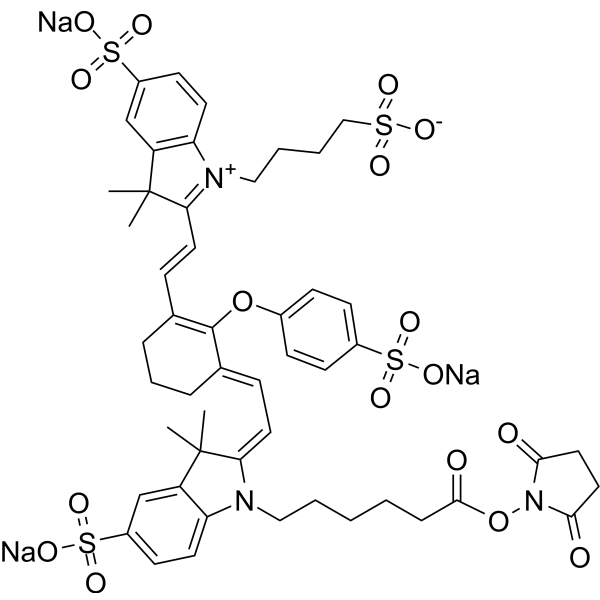
| DC68111 | IRDye 800 NHS Featured |
800CW NHS ester is a near-infrared fluorescent dye active ester that can conjugate with amine-containing small molecules, antibodies, peptides, and other substances to form fluorescent conjugates. 800CW NHS ester exhibits high fluorescence quantum yield and low non-specific binding properties. 800CW NHS ester can be used in biomedical research such as microbial infection imaging, tumor imaging, and biochemical detection.
More description
|
|
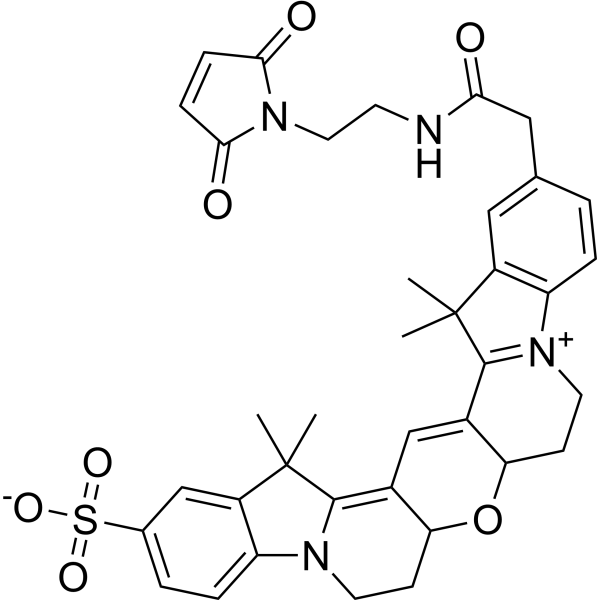
| DC68110 | Cy3B maleimide Featured |
Cy3B maleimide is a mono-reactive dye containing maleimide group, which can selectively and efficiently attach Cyanine3 fluorophore (an analog of Cy3) to proteins and peptides containing cysteine residues, as well as to other thiolated molecules (such as thiol-containing oligonucleotides). Cy3B is an improved version of Cy3 dyes with significantly increased fluorescence quantum yield and photostability.
More description
|
|
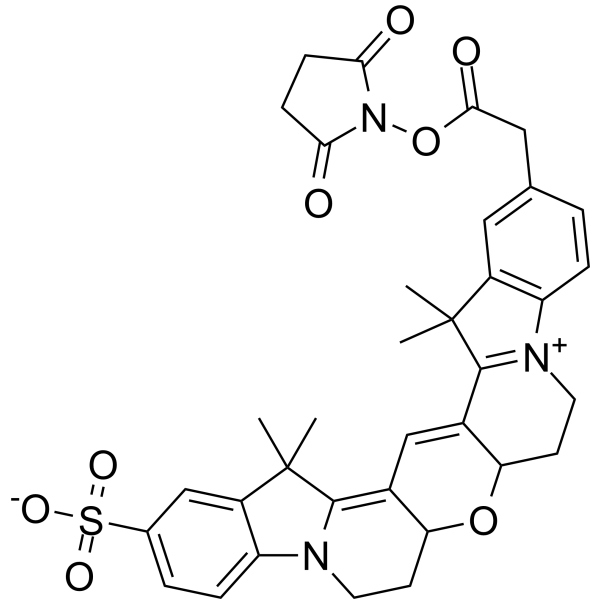
| DC68109 | Cy3B NHS ester Featured |
Cy3B NHS ester is a fluorescent dye compound that is commonly used in biomarking and fluorescent labeling experiments, especially for labeling biomolecules containing amino functional groups (amine groups), such as proteins, antibodies or peptides.
More description
|
|
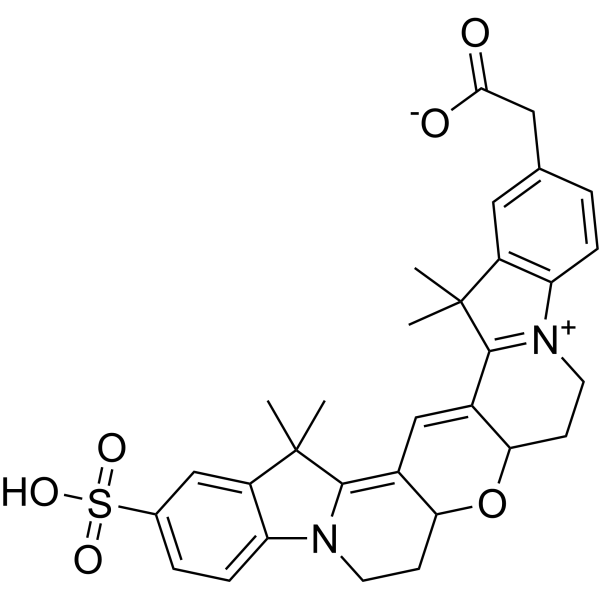
| DC68108 | Cy3B carboxylic acid Featured |
Cy3B is an improved variant of the Cy3 (Cyanine3) (HY-D0822) dye. Cy3 is a fluorescent dye, and its fluorescence spectrum generally falls within the green to orange wavelength range.
More description
|
|
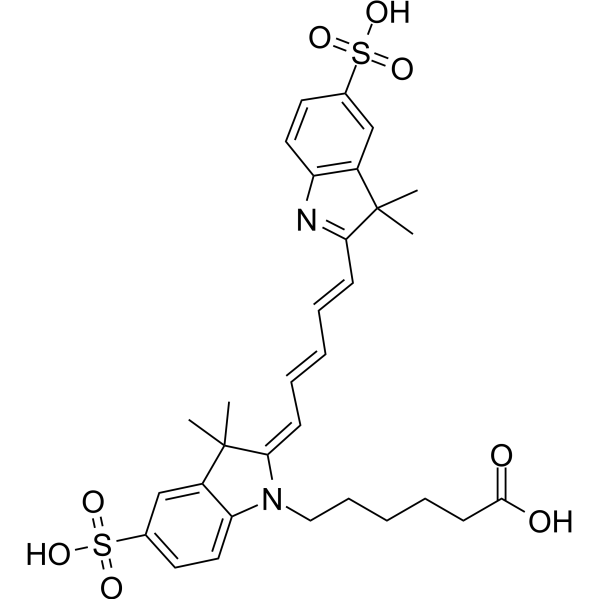
| DC68107 | CypHer5 Featured |
pH Receptor Probe is a pH-sensitive cyanine derivative and a environmental sensitive fluorophore. pH Receptor Probe has a pH-sensitive maximal absorption at 644 nm and emission at 664 nm. pH Receptor Probe has been used as a pH sensor of lysosomal compartments.
More description
|
|
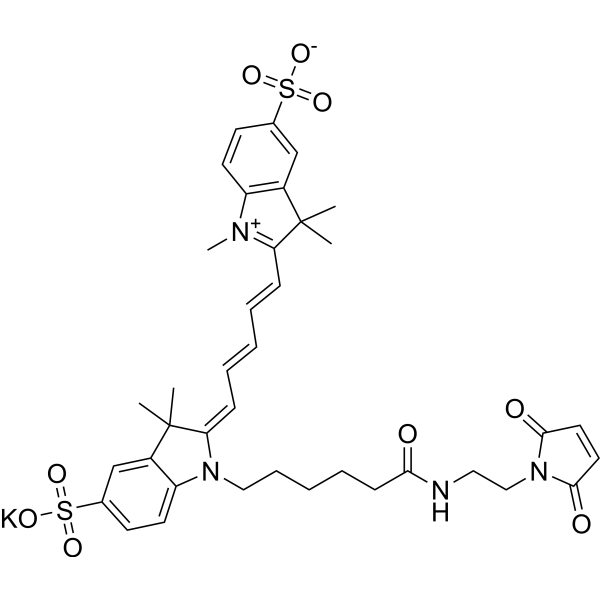
| DC68106 | Sulfo Cy5 Maleimide Featured |
Sulfo-Cy5-Mal potassium is a fluorescent dye derivative composed of a CY5 dye and a maleimide functional group. Sulfo-Cy5-Mal potassium specifically covalently binds to thiol-containing (-SH) biomolecules. Sulfo-Cy5-Mal potassium can be used for protein labeling, antibody conjugation, molecular imaging, and drug delivery studies (Ex/Em = 633 nm/670 nm).
More description
|
|
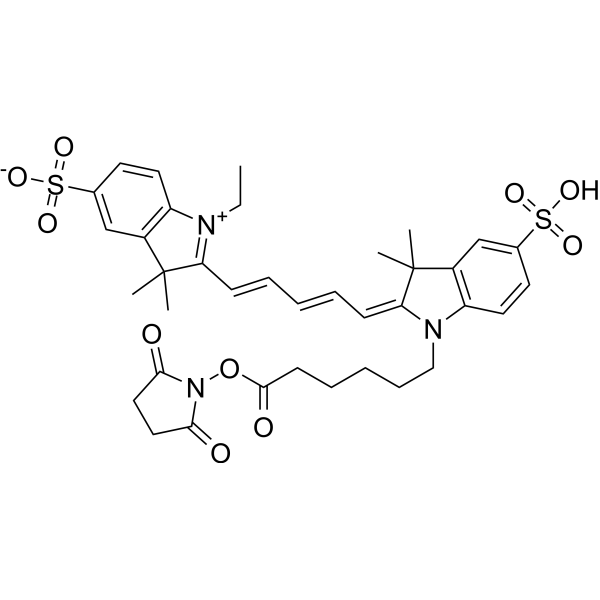
| DC67081 | CY5-SE Featured |
Cy5-SE (Cy5 NHS Ester) is a reactive dye for the labeling of amino-groups in peptides, proteins, and oligonucleotides. This dye requires small amount of organic co-solvent (such as DMF or DMSO) to be used in labeling reaction. This reagent is ideal for very cost-efficient labeling of soluble proteins, as well as all kinds of peptides and oligonucleotides. This reagent also works well in organic solvents for small molecule labeling. Excitation (nm):649, Emission (nm): 670.
More description
|
|
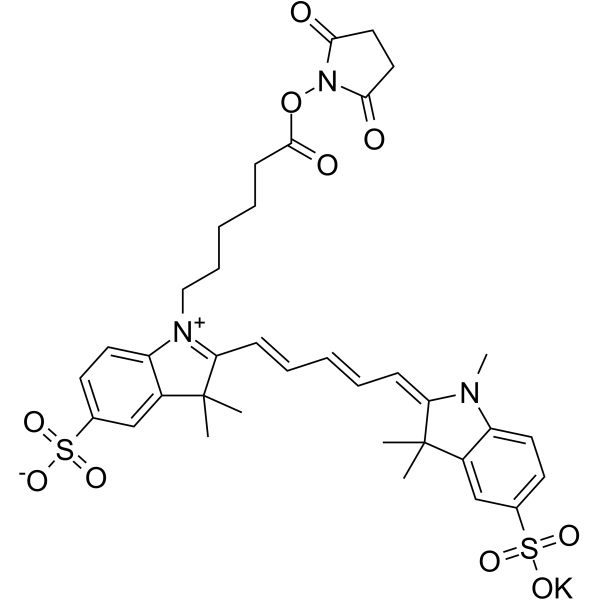
| DC33859 | Sulfo-Cy 5 NHS Ester Featured |
Sulfo-Cyanine 5 NHS Ester is a water soluble amine reactive red emitting fluorescent dye.Used for the labeling of various amine containing molecules in aqueous phase without use of any organic co-solvent. Useful for the labeling of proteins which denature in the presence of organic co-solvents, as well as for proteins with low solubility.
More description
|
|
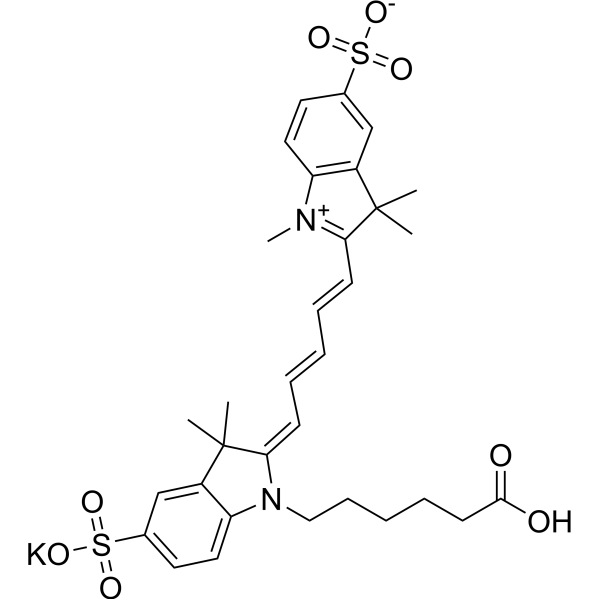
| DC68105 | Sulfo-Cyanine5 carboxylic acid Featured |
Sulfo-Cy5 carboxylic acid potassium is a water-soluble, inactive Cy5 carboxylic acid.
More description
|
|
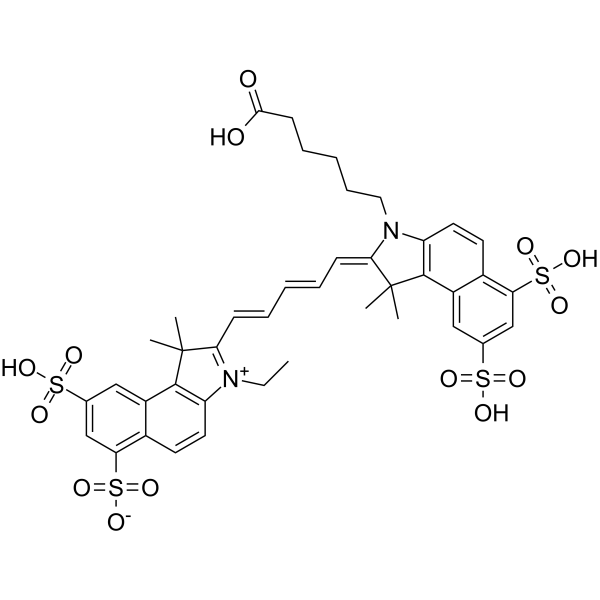
| DC33958 | Sulfo-Cy5.5 acid Featured |
Sulfo-Cyanine5.5 is a water soluble cyanine dye for far red / NIR applications such as in vivo imaging. The dye possesses four sulfonate groups that render it highly hydrophilic and water soluble. As well as other cyanines, sulfo-Cyanine5.5 has an outstanding extinction coefficient that makes it a bright fluorescent label for the far red region.
This is an amine-containing fluorescent dye. The amine group is separated from the fluorophore by a relatively long linker that facilitates conjugation. The aliphatic primary amine group can be coupled with various electrophiles (activated esters, epoxides, etc), and also be used in enzymatic transamination labeling.
More description
|
|
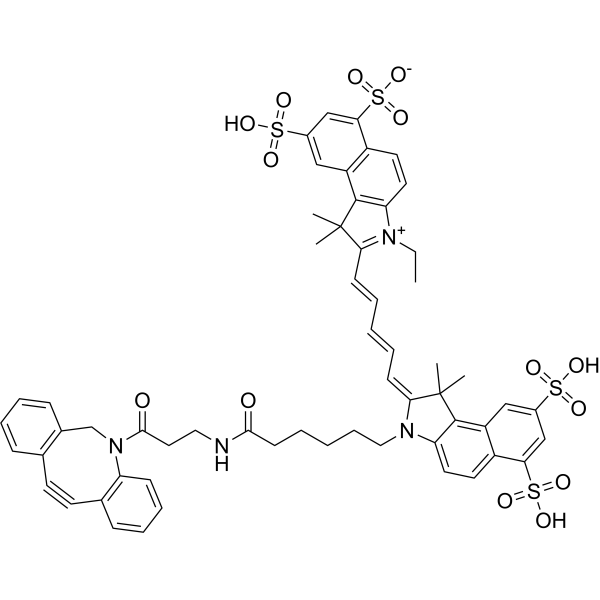
| DC68104 | Cy5.5 DBCO Featured |
Cy5.5 DBCO is a click chemistry reagent containing an cycloalkynes group. Cy5.5 DBCO is a linker of Cyanine5.5 fluorophore. DBCO group enables copper free biocompatible click chemistry with fast reaction kinetics and good stability.
More description
|
|
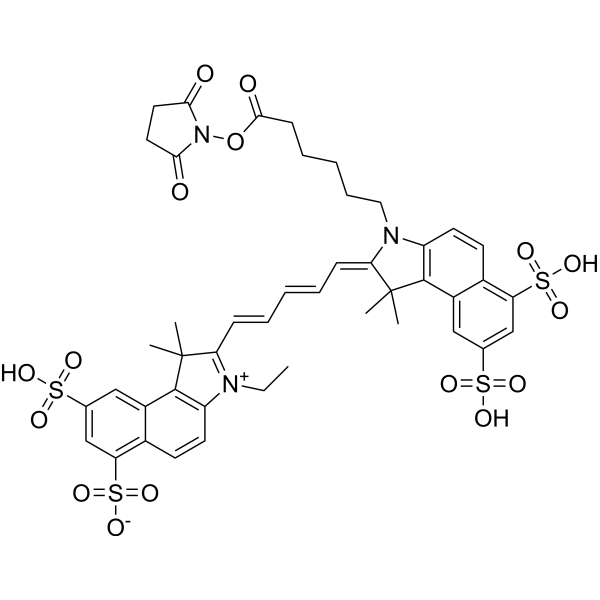
| DC68103 | Cy5.5-SE Featured |
Cy5.5-SE (Cyanine5.5 NHS ester) is a CY dye. CY, short for Cyanine, is a compound consisting of two nitrogen atoms connected by an odd number of methyl units. Cyanine compounds have the characteristics of long wavelength, adjustable absorption and emission, high extinction coefficient, good water solubility and relatively simple synthesis[1]. CY dyes are of en used for the labeling of proteins, antibodies and small molecular compounds. For the labeling of protein antibodies, the combination can be completed through a simple mixing reaction. Below, we introduce the labeling method of protein antibody labeling, which has certain reference significance[2]. Storage: protect from light.
More description
|
|
| DC33885 | Cy5-YNE Featured |
Water soluble reagent with alkyne group for Click Chemistry. Sulfo-Cyanine5 is a sulfonated dye, which possesses high hydrophilicity and aqueous solubility, exceptionally high extinction coefficient, good quantum yield, and compatibility with many instruments.
This reagent is recommended for the conjugation with proteins, nanoparticles, and other applications where hydrophilicity is important.
More description
|
|