 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
| Cat. No. | Product Name | Field of Application | Chemical Structure |
|---|---|---|---|
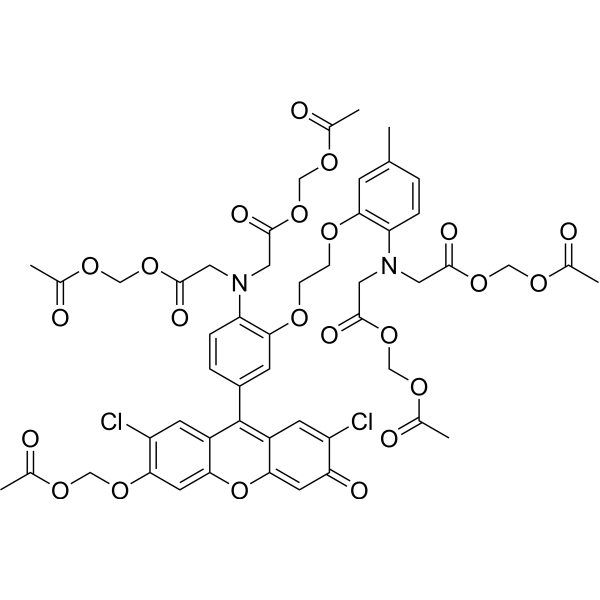
| DCC2137 | Fluo-3 Am Featured |
Fluo-3 AM is a fluorecent Ca2+ chelator, with high affinity for calcium. Fluo-3 AM can specifically identify intracellular calcium ions, with high sensitivity, low cytotoxicity, increased AM acetylmethyl ester can enter the cell well, after being sheared by the intracellular esterase stay in the cell to bind to calcium ions, produce strong fluorescence.
More description
|
|
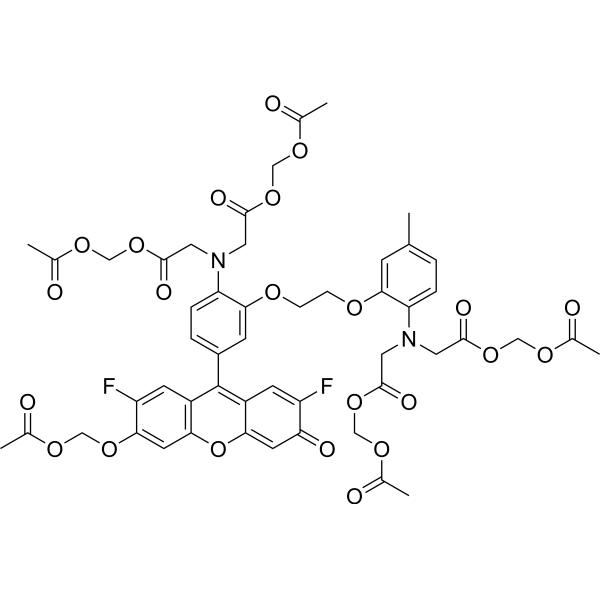
| DC36845 | Fluo-4 AM Featured |
Fluo-4 AM is a cell-permeable, fluorescent Ca2+ indicator.
More description
|
|
| DC71083 | MitoTracker Green FM Featured |
MitoTracker Green FM is a green-fluorescent dye that can selectively accumulate in the mitochondrial matrix. MitoTracker Green FM covalently binds to mitochondrial proteins by reacting with free thiol groups of cysteine residues.
More description
|
|
| DC68130 | MitoTracker Orange CMTMRos Featured |
MitoTracker Orange CMTMRos is a fluorescent dye that labels mitochondria within live cells utilizing the mitochondrial membrane potential (Ex/Em: 551/576 nm).
More description
|
|
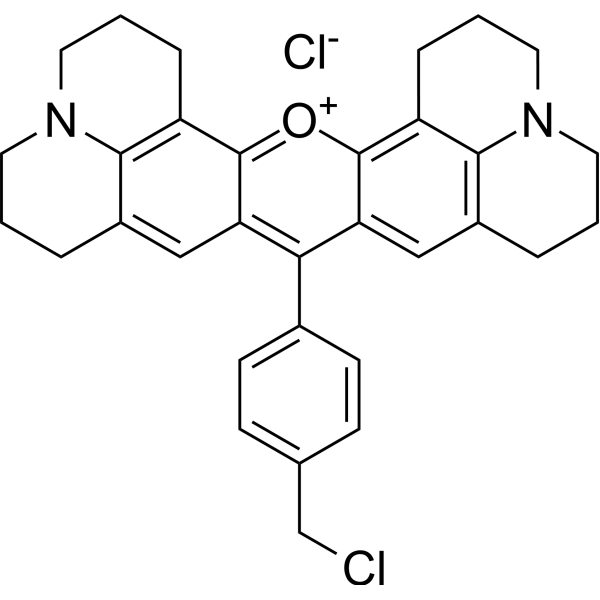
| DC41287 | MitoMark Red I Featured |
MitoMark Red I is a red fluorescent mitochondrial stain. Fluorescence intensity is dependent on mitochondrial membrane potential. Excitation/emission maxima λ ~ 578/599 nm.
More description
|
|
| DC49846 | ER-Tracker Red Featured |
ER-Tracker Red is cell-permeant, live-cell stain that is highly selective for the endoplasmic reticulum (ER).
More description
|
|
| DC45358 | ER-Tracker Green Featured |
ER-Tracker Green is a fluorescent dye that specific for endoplasmic reticulum.
More description
|
|
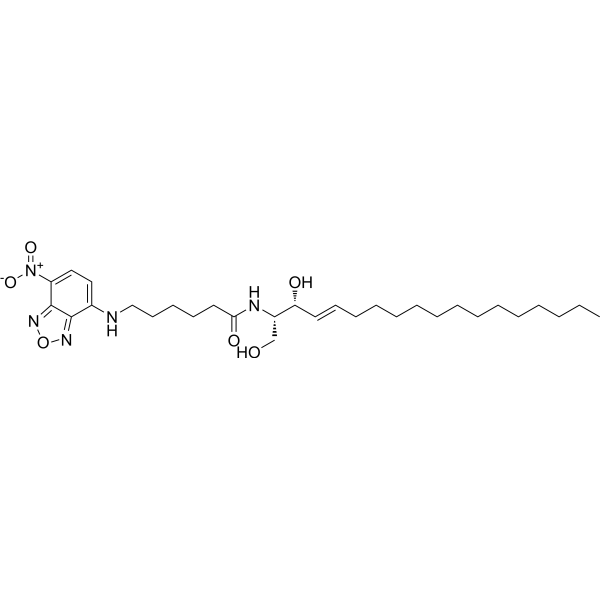
| DC68129 | C6 NBD Ceramide Featured |
C6 NBD Ceramide is a Golgi apparatus fluorescent probe with cell membrane permeability. C6 NBD Ceramide can be used for fast and convenient green fluorescent labeling of Golgi in living and fixed cells, and can be used to observe changes in Golgi morphology in living cells (Ex=466 nm, Em=536 nm). C6-NBD-ceramide is metabolized to fluorescent sphingomyelin and glucosylceramide, can be used for the study of sphingolipid transport and metabolic mechanism.
More description
|
|
| DC68128 | Golgi-Tracker Red Featured |
The Golgi apparatus is composed of flattened vesicles superimposed on each other by unit membranes. The flattened vesicles are round with expanded and perforated edges. The Golgi fluorescent probe is a BODIPY-labeled ceramide derivative, the synthesis of which occurs in the endoplasmic reticulum and can then be transported to the Golgi via ceramide transport protein (CERT) or vesicular translocation, allowing specific labeling of the dye. BODIPY TR Ceramide (Golgi-Red Tracke) is a Golgi-specific fluorescent dye, which can visualise individual cells[1]. Ex/Em=589 nm/616 nm.
More description
|
|
| DC68127 | Golgi-Tracker Green Featured |
The Golgi apparatus is composed of flattened vesicles superimposed on each other by unit membranes. The flattened vesicles are round with expanded and perforated edges. The Golgi fluorescent probe is a BODIPY-labeled ceramide derivative, the synthesis of which occurs in the endoplasmic reticulum and can then be transported to the Golgi via ceramide transport protein (CERT) or vesicular translocation, allowing specific labeling of the dye. BODIPY Fl C5-Ceramide is a Golgi-specific green fluorescent dye, which can visualise individual cells. Ex/Em= 505 nm/512 nm.
More description
|
|
| DC47779 | LysoTracker Red Featured |
LysoTracker Red (LysoTracker Red DND-99) is a paraformaldehyde fixable probe that concentrates into acidic compartments of cells and tissues. LysoTracker Red is an indicator of cell death in a manner similar to other standard assays (Annexin, terminal dUTP nick end labeling, Nile blue sulfate, neutral red, and acridine orange). LysoTracker Red is a marker for late endosomes and lysosomes.
More description
|

|
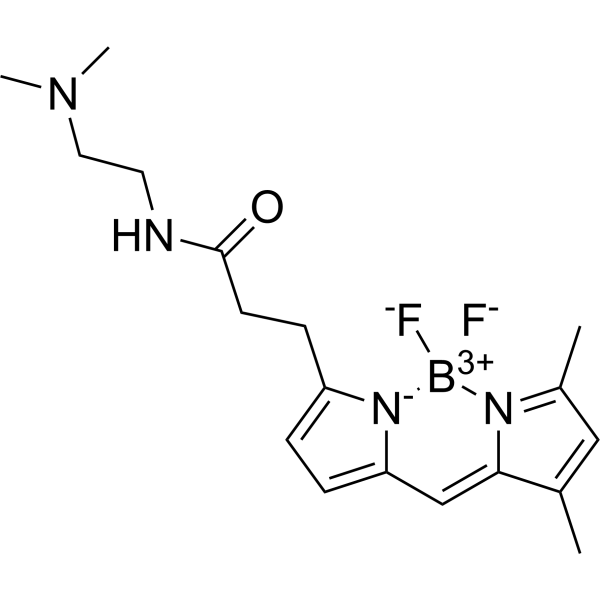
| DC68126 | Green DND-26 Featured |
Green DND-26 is a green fluorescently labeled lysosomal probe with a maximum excitation/emission wavelength of 504/511 nm. The structure is composed of a fluorescein group and linked weak bases, which can freely cross the cell membrane and generally gather on spherical organelles. Green DND-26 is suitable for observing the internal biosynthesis and related pathogenesis of lysosomes.
More description
|
|
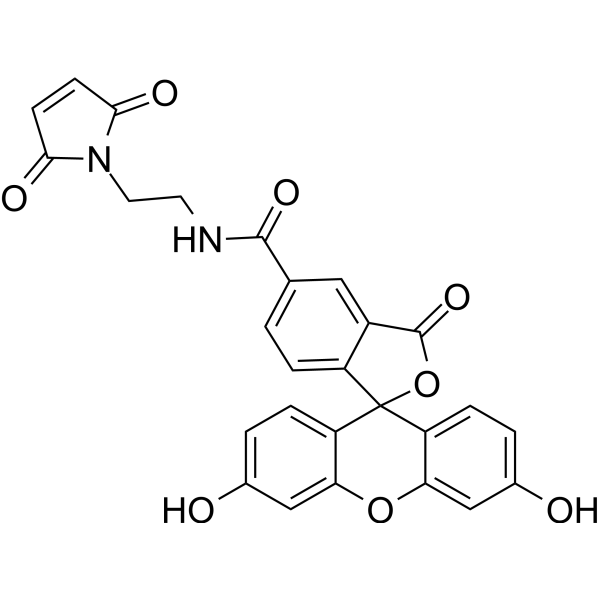
| DC68125 | 5-FAM Maleimide Featured |
Maleimide-5’FAM is a fluorescein. Maleimide-5’FAM can be used for labeling non-transduced cells.
More description
|
|
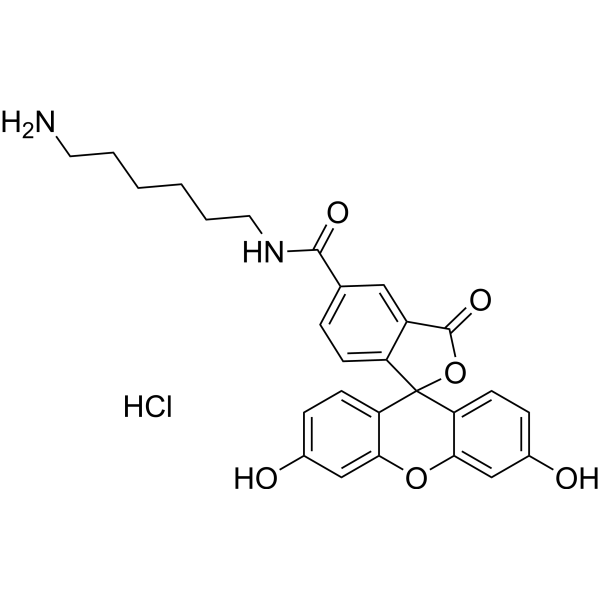
| DC33954 | 5-FAM amine HCl Featured |
Fluorescein (FAM) amine, isomerically pure dye (5-isomer). Unlike fluoresceinamine, this reagent contains a linker arm with an aliphatic amine group that is more reactive. It can react with electrophilic reagents like activated esters or epoxides, and be involved in enzymatic transamination.
More description
|
|
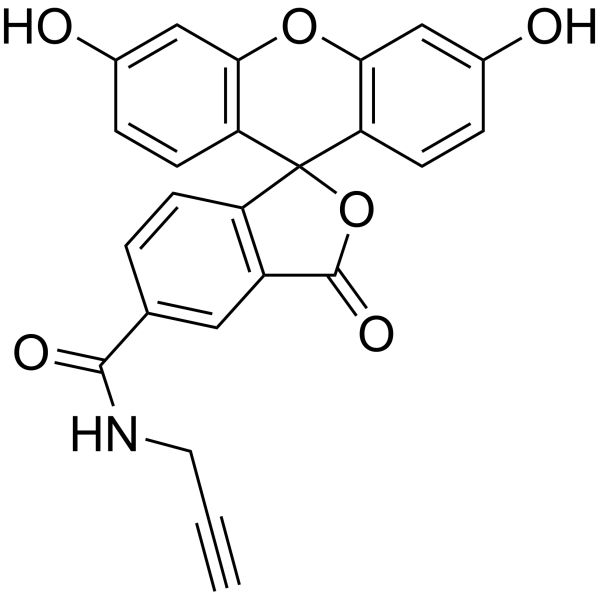
| DC68123 | 5-FAM alkyne Featured |
5-FAM-Alkyne is a high selective and sensitive fluorescent biosensor for alkaline phosphatase (ALP). 5-FAM-Alkyne is a click chemistry reagent, it contains an Alkyne group and can undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups.
More description
|
|
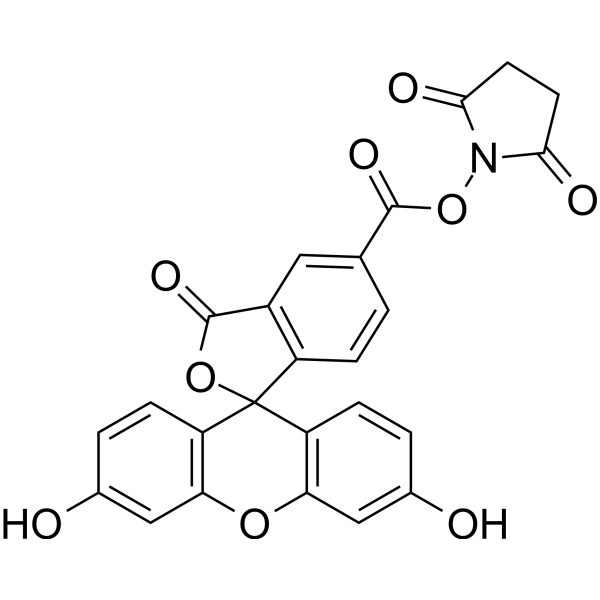
| DC9371 | 5-FAM SE Featured |
5-FAM SE is a single isomer, it is one of the most popular green fluorescent reagents used for labeling peptides, proteins and nucleotides.
More description
|
|
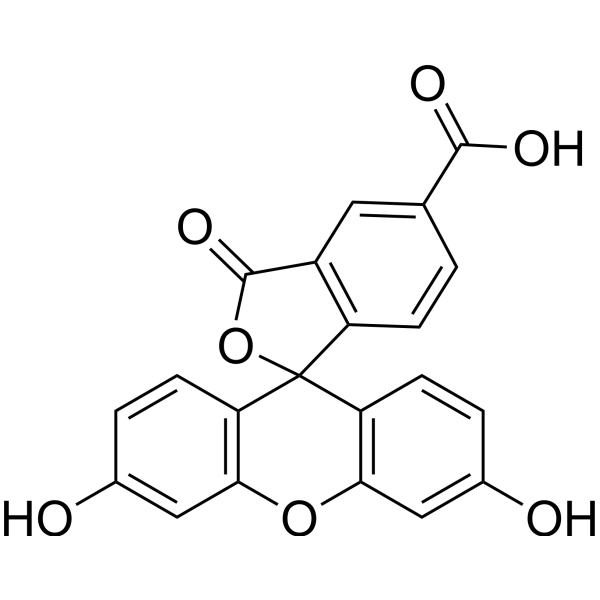
| DC22480 | 5-FAM Featured |
The single isomer, 5-FAM, contains a carboxylic acid that can be used to react with primary amines via carbodiimide activation of the carboxylic acid. Fluorescein is the most common fluorescent derivatization reagent for labeling biomolecules. In addition
More description
|
|
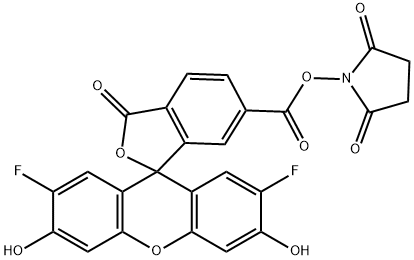
| DC68122 | 6-OG488 NHS ester Featured |
|
|
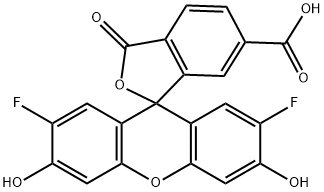
| DC68121 | 6-OG488 acid Featured |
|
|
| DC74921 | OG-488 SE Featured |
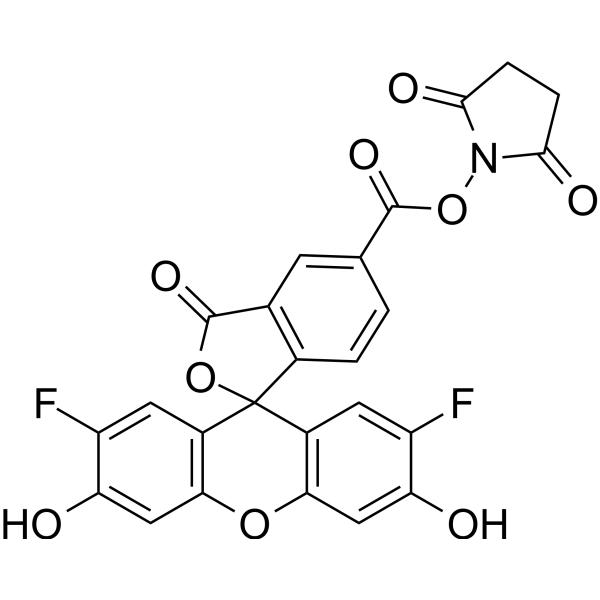
OG 488 SE is a Green fluorescent dye; supplied as NHS ester. OG-488 SE is a potential fluorescein precursor for live bacteria detection.
More description
|
|
| DC74549 | EOS789 Featured |
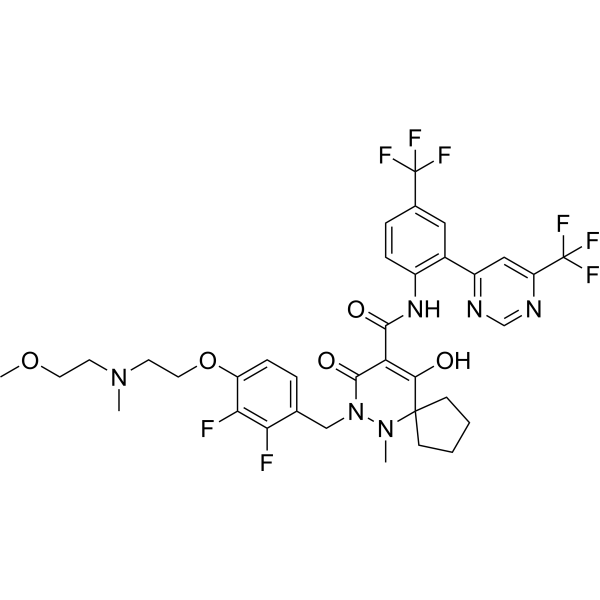
EOS789 is a potent, pan-phosphate transporter inhibitor, inhibits phosphate uptake in a noncompetitive manner for human NaPi-IIb, PiT-1, and PiT-2 with IC50 of 6.8, 1.5 and 1.7 uM, respectively.
More description
|
|
| DC47813 | OG 488, acid Featured |
OG 488, acid, a fluorescent pH indicator, has many applications in biochemistry and neurosciences.
More description
|

|
| DC68120 | 2',7'-Difluorofluorescein Featured |
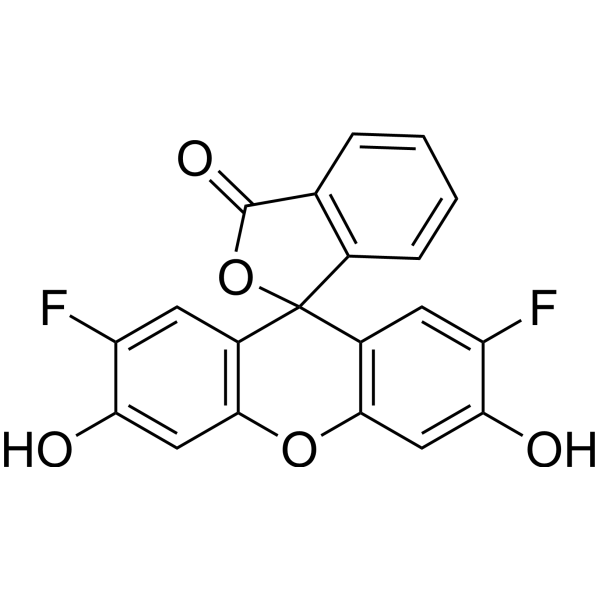
2',7'-Difluorofluorescein (Oregon green 488) is a fluorescein derivative and a pH-sensitive fluorescent probe (pKa ~4.7). Upon excitation at 488 nm, 2',7'-Difluorofluorescein exhibits pH-sensitive fluorescence intensity through the formation of dianions, while its pH sensitivity decreases under excitation at 450 nm, allowing its use in ratiometric pH analysis. 2',7'-Difluorofluorescein can be used for the quantitative analysis of pH values in the range of 2-7 in submicron aerosol particles.
More description
|
|
| DC68119 | Texas Red Featured |
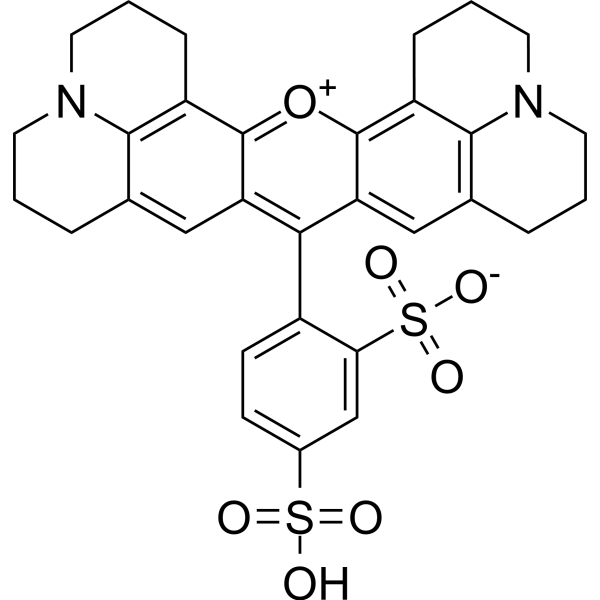
Texas Red (Sulforhodamine 101) is an amphoteric rhodamine red fluorescent dye (excitation/emission: 586/605 nm). Texas Red is used extensively for investigating neuronal morphology and acts as acell type-selective fluorescent marker of astrocytes bothin vivoand in slice preparations.
More description
|
|
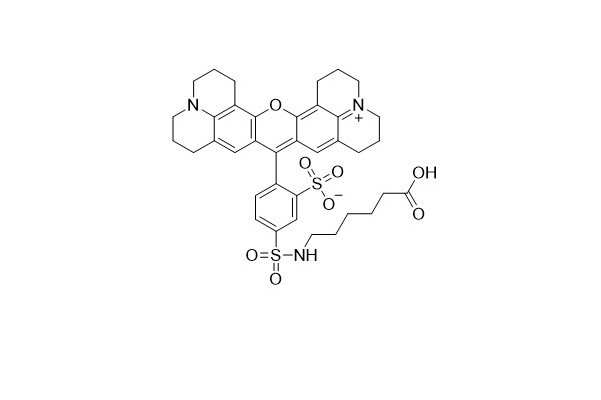
| DC67959 | Tex Red-X Acid Featured |
Tex Red-X dye is an innovative alternative to Tex Red. A key difference lies in its chemical structure - it features an additional seven - atom aminocaproyl spacer ("X") between the fluorophore and the carboxyl group. This spacer plays a crucial role in separating the fluorophore from its attachment point. By doing so, it effectively minimizes the interaction between the fluorophore and the conjugated biomolecule.
One of the standout features of Tex Red - X is that it exists as a single 5 - isomer. This characteristic significantly enhances the reproducibility of results, which is a highly desirable trait in scientific research and biological applications.
In terms of optical properties, Tex Red - X shares the same spectral characteristics as Texas Red. It can be utilized to create bright red fluorescent bioconjugates with excitation and emission maxima at approximately 595/615 nm. These bioconjugates are valuable tools in various biological and biochemical studies, enabling researchers to visualize and analyze biological processes with high precision.
More description
|
|
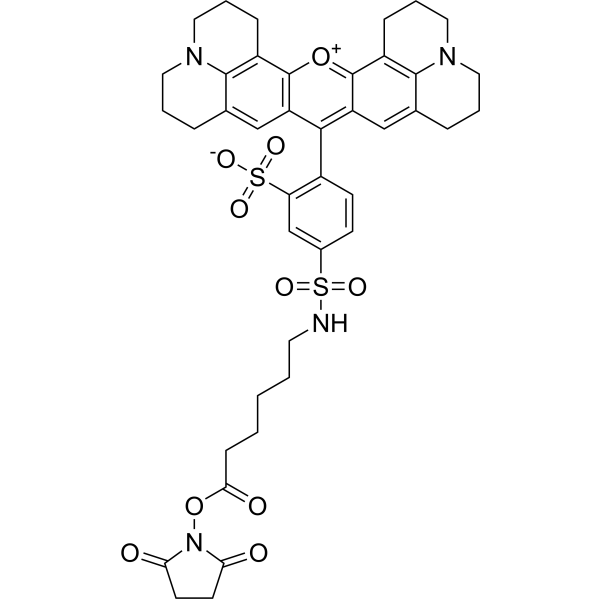
| DC67951 | Tex Red-X NHS Featured |
Tex Red-X NHS is a derivative of Texas Red (HY-101878), an amphoteric rhodamine red fluorescent dye. Texas Red is widely used to study neuronal morphology and as a cell type-selective fluorescent marker for astrocytes, both in vivo and in slice preparations.
More description
|
|
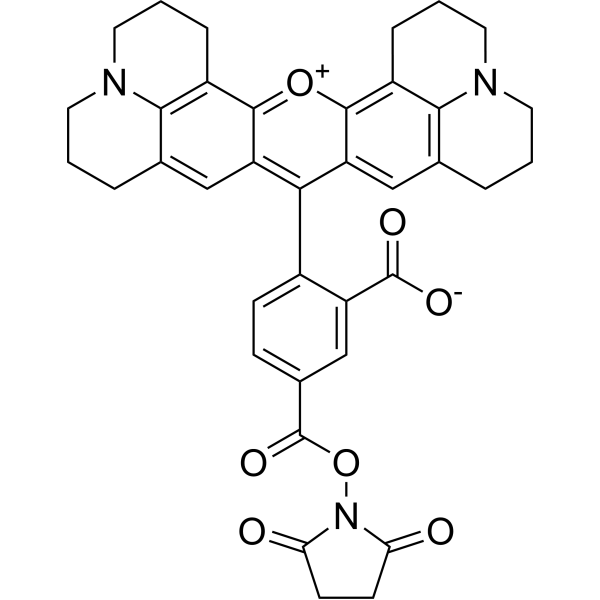
| DC68118 | 5-ROX NHS ester Featured |
5-Carboxy-X-rhodamin N-succinimidyl ester (5-ROX, SE) is a labeling reagent. 5-Carboxy-X-rhodamin N-succinimidyl ester can be used in the preparation of charge-modified dye-labeled Dideoxynucleotide Triphosphates (ddNTP) to "direct-load" DNA sequencing.
More description
|
|
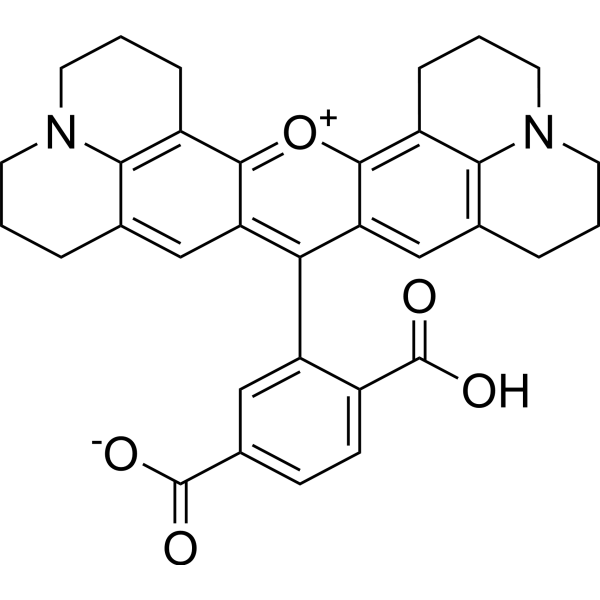
| DC60916 | 6-ROX Featured |
6-ROX is a selective fluorescent probe and potential inhibitor of COX-2. 6-ROX binds to the active site of COX-2 and inhibits its conversion of arachidonic acid into prostaglandins. 6-ROX is often used in the field of optical imaging related to tumors and inflammation, and helps detect diseased tissues with high expression of COX-2.
More description
|
|
| DC67949 | 6-ROX NHS Featured |
6-Carboxy-X-rhodamine, succinimidyl ester (6-ROX, SE) is a fluorescent dye for oligonucleotide labeling and automated DNA sequencing.
More description
|
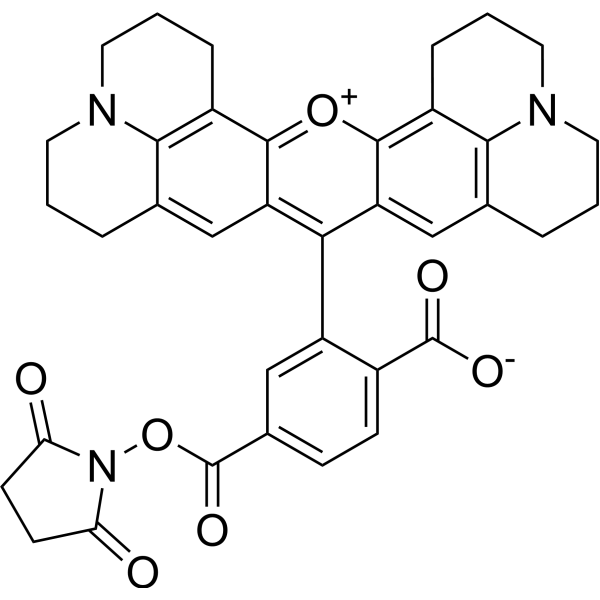
|
| DC70522 | JMN3-003 Featured |
JMN3-003 is a host-directed inhibitor with potent antiviral activity against a panel of myxovirus family members with EC50 of 10-70 nM.JMN3-003 shows activity against MeV at 170 (viral CPE-reduction assay) and 30 nM (virus yield reduction assay) and does not display any detectable acute cytotoxicity.JMN3-003 also shows superb antiviral activity against a selection of clinical-relevant paramyxovirus (RSV, MuV, and HPIV3) and orthomyxovirus (influenza) family members.
More description
|
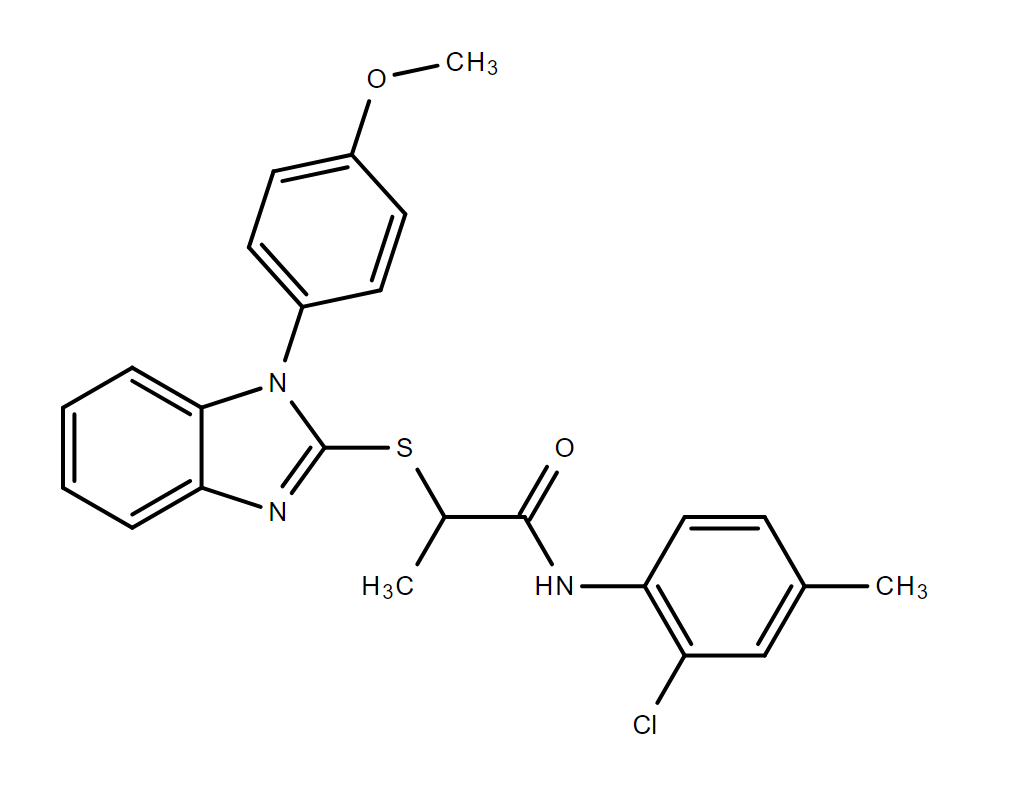
|