 To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
To enhance service speed and avoid tariff delays, we've opened a US warehouse. All US orders ship directly from our US facility.
| Cat. No. | Product Name | Field of Application | Chemical Structure |
|---|---|---|---|
| DC65329 | ALC-0315 analogue-2 Featured |
ALC-0315 analogue-2 is an analogue of ALC-0315. ALC-0315 is an ionisable aminolipid that is responsible for mRNA compaction and aids mRNA cellular delivery and its cytoplasmic release through suspected endosomal destabilization. ALC-0315 can be used to form lipid nanoparticle (LNP) delivery vehicles. Lipid-Nanoparticles have been used in the research of mRNA COVID-19 vaccine.
More description
|

|
| DC65434 | SM102 Analog 1 Featured |
An analog of SM-102. The ethanolamine amino lipid head enhances encapsulation of mRNA. The lipid has primary esters at C7 position relative to the amine nitrogen. The primary lipid tail has 8 carbon tail. The lipid can be used for mRNA-based therapies which depends on the availability of a safe and efficient delivery vehicle.
More description
|

|
| DC65334 | Lipid 15 Featured |
Lipid 15 is an ionizable amino lipid used for the generation of Lipid nanoparticles .
More description
|

|
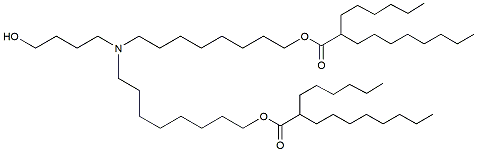
| DC65349 | ALC-0315 analgous-3 Featured |
ALC-0315 analgous-3 is an butanolamine ionizable lipid with both ester bonds located adjacent to C8 relative to the amine head. The introduction of ester linkages can improve the clearance of the lipid in the liver. This compound is analgous to ALC-0315.
More description
|
|
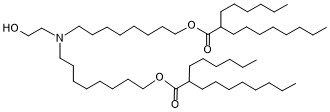
| DC83220 | ALC-0315 analogous-1 Featured |
ALC-0315 analogous-1 is a derivative of the ionizable cationic amino lipid ALC-0315. It has been used in the synthesis of ionizable cationic lipids used in the generation of lipid nanoparticles (LNPs).
More description
|
|
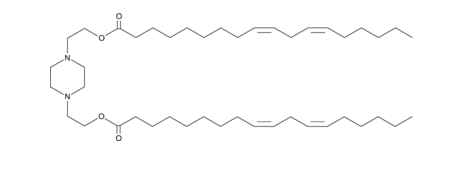
| DC82101 | AA3-DLin Featured |
AA3-DLin is an ionizable cationic amino lipid (pKa = 5.8) that has been used in combination with other lipids in the formation of lipid nanoparticles (LNPs) for the delivery of mRNA.LNPs containing AA3-DLin and encapsulating mRNA for the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike glycoprotein induce immunogenicity in mice.
More description
|
|
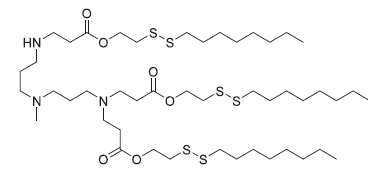
| DC82025 | 306-O12B-3 Featured |
306-O12B-3 is an ionizable lipidoid with cationic properties, commonly used in lipid nanoparticle (LNP) formulations for antisense oligonucleotide (ASO) delivery. When administered intravenously in mice, LNPs incorporating 306-O12B-3 exhibit liver-specific accumulation. Studies show that ASO-loaded LNPs containing 306-O12B-3 effectively silence hepatic PCSK9 expression by targeting the proprotein convertase subtilisin/kexin type 9 gene. Additionally, when combined with the cationic lipidoid NT1-O14B (Item No. 37095), these LNPs can deliver tau-targeting ASOs to the brain, reducing tau protein levels in mice.
More description
|
|
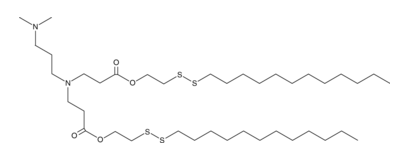
| DC82305 | 80-O16B Featured |
80-O16B is a disulfide bond-containing ionizable cationic lipidoid. It has been used in the generation of lipid nanoparticles (LNPs) for the delivery of CRISPR complementary single-guide RNA (sgRNA) and Cas9 for genome editing in mice. LNPs containing 80-O16B conjugated to phenylboronic acid (PBA) and encapsulating an mRNA reporter increase luciferase reporter expression in HeLa cancer cells.2 LNPs containing 80-O16B conjugated to PBA and encapsulating p53 mRNA decrease the viability of DU145 prostate and SiHa and HeLa cervical cancer cells.
More description
|
|
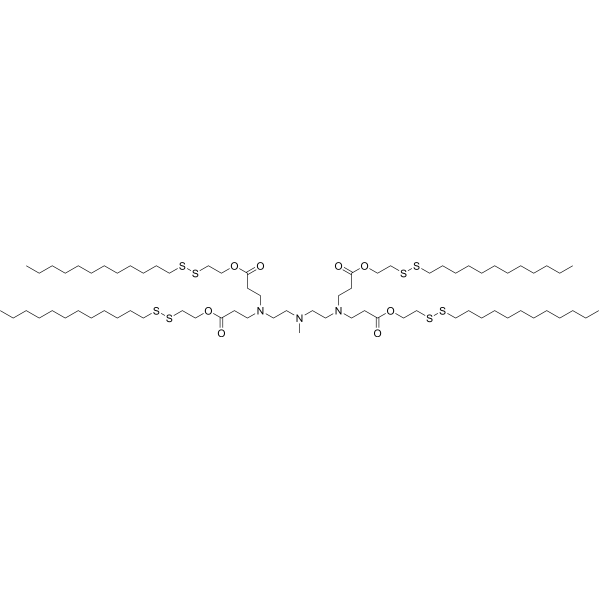
| DC82119 | 113-O16B Featured |
113-O16B is a disulfide bond-containing ionizable cationic lipidoid. It has been used in the generation of lipid nanoparticles (LNPs) for the delivery of mRNA.
More description
|
|
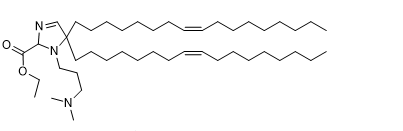
| DC82003 | A12-Iso5-2DC18 |
A12-Iso5-2DC18 is a novel amine containing lipid can be used for mRNA delivery, activate the stimulator of interferon genes (STING) pathway, and exhibit anti-tumor immunity.
More description
|
|
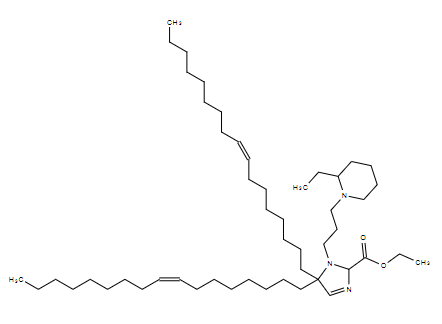
| DC80071 | A18-Iso5-2DC18 Featured |
A18-Iso5-2DC18 that could not only deliver mRNA vaccines robustly but also activate the stimulator
of interferon genes (STING) pathway. A18-Iso5-2DC18 strongly binds to the stimulator of interferon genes (STING) and induces potent cytolytic T lymphocyte responses, resulting in substantial antitumor immunity (Miao et al. 2019).
More description
|
|
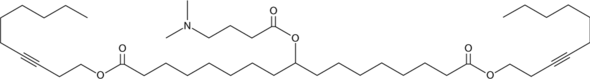
| DC80050 | LIPID A6 Featured |
Lipid A6 is an ionizable cationic and biodegradable alkyne lipid (pKa = 6.65).It has been used with other lipids in the formation of lipid nanoparticles (LNPs) for the delivery of mRNA. LNPs containing lipid A6 and encapsulating mRNA encoding human erythropoietin (EPO) increase and then maintain homeostatic levels of hemoglobin in the blood in an adenine-induced mouse model of renal anemia.
More description
|
|
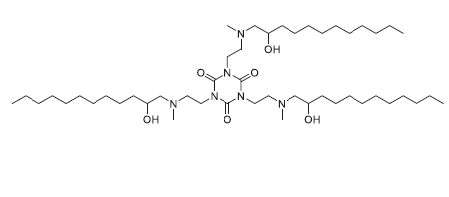
| DC83230 | TNT-b10 Featured |
TNT-b10 is a novel Lipid-like compound suitable for delivery of siRNA and mRNA both in vitro and in vivo TNT-b10 LLNs was more than 10-fold more potent than TNT-a10 LLNs formulated under the same condition.
More description
|
|
| DC65701 | L-369 Featured |
L-369 (Lipid 369,L369) is novel class of ionizable lipid for siRNA delivery with improved in vivo elimination profile with excellent translation across species,including NHP, wide safety margin.
More description
|

|
| DC49889 | 503O13 Featured |
503O13 is a next-generation, biodegradable lipid nanoparticle (LNP) engineered for highly efficient and targeted siRNA delivery. Designed through rational structure-activity criteria—including optimal tail length (O13), tertiary amines, and a surface pKa ≥5.5—this single-component LNP achieves unparalleled gene silencing with an ultra-low EC50 of 0.01 mg/kg in preclinical models.503O13 outperforms non-degradable counterparts (e.g., C12-200) with improved toxicity profiles—no hepatic necrosis or pancreatic inflammation—while maintaining rapid blood clearance (t1/2: 6 min) and organ-specific accumulation (liver/spleen).
More description
|

|
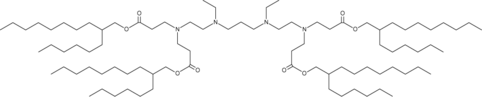
| DC60546 | 514O6,10 Featured |
514O6,10 is an ionizable lipidoid. 514O6,10 formulated LNPs facilitate mRNA delivery to the pancreas.
More description
|
|
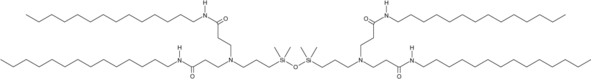
| DC60663 | Si5-N14 Featured |
Si5-N14 is a lipid-based molecule engineered with siloxane groups, designed specifically for efficient mRNA delivery to the lungs. The incorporation of siloxane units boosts the cellular uptake of mRNA-loaded lipid nanoparticles (LNPs) and enhances their ability to escape from endosomes. These properties significantly increase the overall effectiveness of mRNA delivery, making Si5-N14 a promising tool for targeted therapeutic applications.
More description
|
|
| DC67515 | CICL-207 Featured |
CICL 207 is structurally optimized based on Lipid CICL-1. CICL207 is a constrained ionizable cationic lipid designed for lipid nanoparticle (LNP) delivery systems developed by Capstan. Its structure features a rigid cyclic backbone (e.g., pyrrolidine-derived core) paired with a tertiary amine group that ionizes at acidic pH (pKa ~6.5–7.0), enhancing endosomal escape. The lipid includes asymmetric hydrophobic tails (likely C14–C18 alkyl/ester chains) to stabilize LNP membranes and improve nucleic acid encapsulation. Integrated into LNPs (e.g., 58% CICL-207, 10% DSPC, 30.5% cholesterol, PEG-lipids), it enables targeted delivery to T cells (anti-CD5/CD8 tLNPs) with high transfection efficiency (spleen T cells >70% mCherry+), reduced liver uptake, and low toxicity (no significant ALT/AST elevation in rats). Its constrained design balances stability, tissue specificity, and biocompatibility for gene therapy applications.CICL 207 (F50) significantly outperforms CICL-1 by delivering dramatically enhanced target cell transfection with reduced off-target effects. It achieves >50% transfection efficiency in splenic T-cells—nearly double that of CICL-1—while slashing off-target expression in liver cells to <5% (versus >15% for CICL-1. This precision translates to superior therapeutic outcomes: CICL-207 enables ~95% B-cell depletion in CAR-T applications, far exceeding CICL-1 ’s ~60% efficacy. Critically, it maintains an exceptional safety profile, showing no significant liver toxicity or inflammatory cytokine elevation even at high doses. Furthermore, CICL-207 demonstrates 2-fold higher transfection efficiency in hematopoietic stem cells, enabling robust gene editing. Its optimized pKa (~6.5) and constrained amine structure enhance endosomal escape while minimizing Kupffer cell uptake, making it ideal for targeted therapeutics requiring both potency and safety.
More description
|

|
| DC60856 | DMA4-H228 Featured |
DMA4-H228 is a novel, biodegradable lipidoid specifically engineered for spleen-targeted mRNA delivery. Its structure combines a dimethylamino (DMA4) headgroup with a unique hyperbranched lipid tail (H228) synthesized via Michael addition, incorporating ester bonds for enhanced biodegradability. This design enables the formation of stable lipid nanoparticles (LNPs) (~170 nm) with high mRNA encapsulation efficiency (>96%).
Critically, DMA4-H228 exhibits exceptional intrinsic tropism for the spleen (>98% targeting efficiency after IV administration), requiring no external targeting ligands. It selectively delivers mRNA to splenic antigen-presenting cells (APCs), including dendritic cells, macrophages, and B cells. This triggers potent immune activation: rapid IFNα secretion, upregulation of APC maturation markers (CD86/CD40), and robust antigen-specific immune responses.
Demonstrating significant therapeutic potential, DMA4-H228-based mRNA vaccines effectively inhibit tumor growth in melanoma models (e.g., B16F10-OVA). This correlates with increased tumor-infiltrating CD8⁺ T cells, a shift towards pro-inflammatory M1 macrophages, elevated antigen-specific antibodies (IgG), and strong T cell responses (evidenced by IFNγ⁺ spots). Its ability to bypass liver tropism and directly activate splenic APCs makes DMA4-H228 a powerful platform for next-generation mRNA vaccines and cancer immunotherapy.
More description
|

|
| DC67537 | DM3-BTA-14 Featured |
DM3-BTA-14 is a cationic lipid compound engineered for high-efficiency mRNA delivery developed by Hefei AlphaNA Biotechnology. Its structure features a rigid benzene-1,3,5-tricarboxamide core linked to a protonatable dimethylamino headgroup (-N(CH₃)₂) via a propylene spacer (-CH₂CH₂CH₂-) and two saturated C14 alkyl chains. This design enables ≈90% endosomal escape efficiency , superior lymph node targeting for vaccines , and effective tumor-specific mRNA delivery . It outperforms benchmark lipids while maintaining low cytotoxicity, forming stable nanoparticles with cholesterol/DSPC/DSPE-PEG (50:39:10:1 ratio) for therapeutic applications.
More description
|

|
| DC59217 | Arcturus lipid 2(ATX-0114) Featured |
Lipid 2,2(8,8) 4C CH3 is an ionizable cationic lipid (pKa = 6.69).1 It has been used in the generation of lipid nanoparticles (LNPs) for the delivery of siRNA in vivo. LNPs containing lipid 2,2(8,8) 4C CH3 and encapsulating siRNA targeting Factor VII decrease plasma Factor VII protein levels by 90% in mice.
More description
|

|
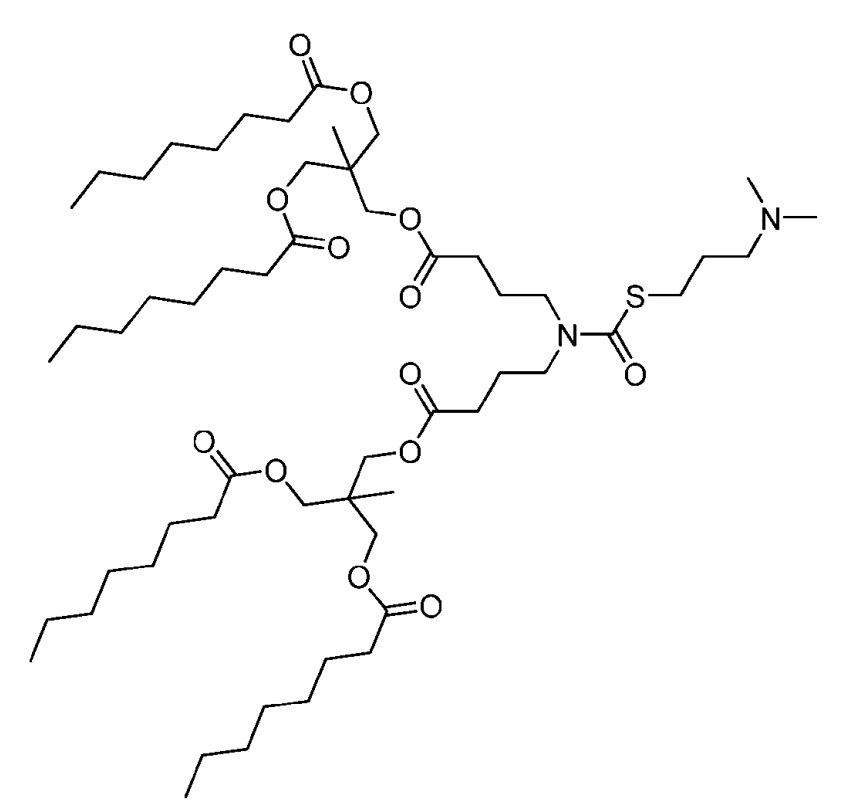
| DC67291 | ATX054 Featured |
ATX-054 which is from Arcturus RNA delivery platform, is a novel ionizable lipid used in the formulation of lipid nanoparticles (LNPs) for the delivery of RNA.
More description
|
|
| DC59126 | Genevant CL1 (lipid 10) Featured |
Genevant CL1 (lipid 10) is a novel ionizable lipid for rna delivery.Lipid 10 rapidly accumulated in the liver within the first hour of dosing (reflecting LNP uptake), but levels then steadily declined over the ensuing 2 weeks period, similar to MC3.Lipid 10 afforded more than double the expression of either approved lipid. We also observed high splenic expression for ALC-0315, which correlated with higher MCP-1 levels.Animals received a single 5 µg IM dose of LNP encapsulating firefly luciferase (fLuc) mRNA. Whole body imaging was performed 6 h later and expression at the injection site quantified. Lipid 10, ALC-0315, and SM-102 showed similar expression at the injection site, all greater than the older generation benchmarks lipids (DLinDMA, KC2, MC3). Lipid 10 and ALC-0315 also showed high expression in the liver, while SM-102 was less, and more similar to MC3.Lipid 10-based LNP reported similar anti-HA IgG titers to MC3 and ALC-0315 (Comirnaty) LNP, and higher than the SM-102 (SpikeVax) LNP composition. MCP-1 levels were generally similar, although the ALC-0315 composition had a significantly higher response at the 5 µg dose. All formulations reported good stability when stored frozen at −80 °C or at 2–8 °C for 1 month.
More description
|

|
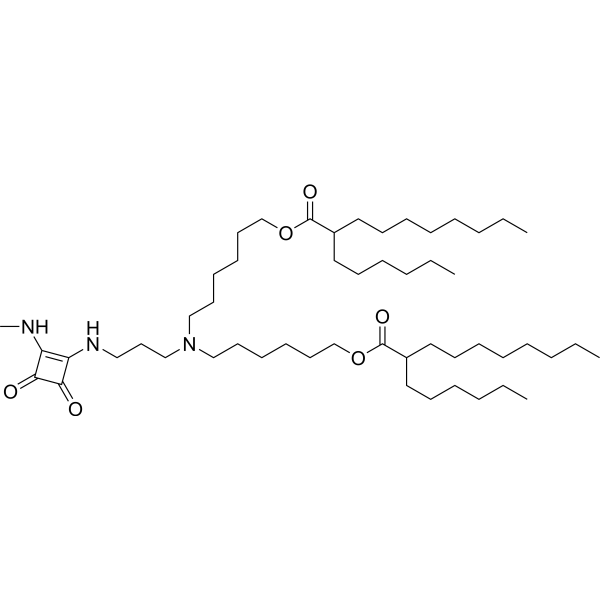
| DC67128 | Lipid 29 analogue-2(Lipid 16) Featured |
Lipid 29 analogue-2 is an ionizable lipid designed for the delivery of RNA-based therapeutics, such as mRNA or siRNA.
More description
|
|
| DC71687 | Dlin-MeOH Featured |
Dlin-MeOH is a lipid product for use in drug delivery systems.
More description
|

|
| DC67563 | S-Ac7-DOg |
S-Ac7-DOg is an ionizable lipid engineered for optimized mRNA delivery to the retina, featuring a sulfur-based ester bond (S-Ac) and dual oleyl glyceride chains (DOg). Its pKa (~6.74) is finely tuned to enhance endosomal escape in acidic environments, enabling efficient cytosolic mRNA release. Unlike traditional lipids (e.g., C12-200, MC3), S-Ac7-DOg incorporates biodegradable ester linkages that hydrolyze intracellularly, minimizing lipid accumulation and reducing innate immune activation.
In vitro, S-Ac7-DOg LNPs achieved >80% transfection efficiency in retinal cells (ARPE-19, MIO-M1) with negligible cytokine secretion, outperforming MC3 and rivaling C12-200 while avoiding the latter’s high immunogenicity. In vivo, intravitreal delivery in mice showed robust protein expression in the optic nerve head (ONH) and Müller glia (75–100% of eyes), sustained for ≥7 days. Critically, it induced the lowest immunogenicity among tested lipids: minimal leukocyte infiltration (<1.5-fold vs. PBS), no microglial reactivity, and reduced GFAP upregulation.
More description
|

|
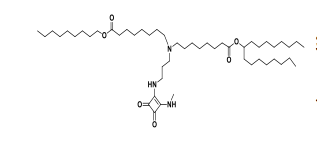
| DC67218 | Moderna Lipid compound 182(Lipid 29 analogue-1) Featured |
Moderna Lipid compound 182(Lipid 29 analogue-1) is a novel ionizable amine lipid developed by Moderna for the delivery of mRNA-based therapeutics. This lipid is part of Moderna's proprietary lipid nanoparticle (LNP) delivery platform, which is designed to encapsulate and protect mRNA, facilitate its cellular uptake, and enable efficient intracellular release. The ionizable nature of Lipid Compound 182 allows it to interact with mRNA at low pH (during LNP formulation) and release the payload in the neutral pH environment of the cytoplasm, making it a critical component of Moderna's mRNA delivery system.
More description
|
|
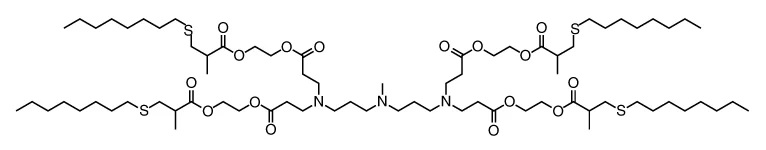
| DC82001 | 4A3-SC8 Featured |
4A3-SC8 is a novel Ionizable amino lipid for RNA delivery.The CRISPR-Cas9 gene editing system has been a hotspot in the
field of gene therapy, especially the gene correction induced by
homology-directed repair (HDR). However, its application has
various obstacles, such as large molecular weight, poor stability,
off-target risk, and the complexity of codeliver multiple genes.
Farbiak et al. established a novel ionizable lipid library consisting
of four distinct amine cores (3A3, 3A5, 4A1, 4A3) and nine
peripheries with different alkyl chain lengths (SC5-SC14), and screened out a class of iLNPs with ability of encapsulating
Cas9 mRNA, sgRNA and donor DNA simultaneously.
The delivery efficiency (quantified by luciferase mRNA expression)
and iLNPs toxicity were evaluated with three different cell
lines (HEK293T, HeLa, and IGROV-1), indicating the formulation
containing 4A3-SC8 was the best. 4A3-SC8 iLNPs successfully
induced HDR in HEK293 cells by one-pot delivery of Cas9
mRNA, sgRNA, and the correct ssDNA template. Confocal
microscopy imaging showed that a portion of blue fluorescence
in cells was corrected to green fluorescence. Furthermore, the
nucleic acid ratios of Cas9: sgRNA: donor DNA loading in
iLNPs at a ratio of 2:1:3 could maximize the HDR efficiency with
the editing efficiency up to 23%, which breaks through the current
bottleneck of HDR efficiency of only 1–5%. This progress is
undoubtedly an important advance in the gene therapy field to
cure diseases caused by genetic mutations.
More description
|
|
| DC67216 | Moderna Lipid 26(Lipid M) Featured |
Moderna Lipid 26(Lipid M) is an ionizable cationic lipid (pKa = 6.75) that has been used in the generation of lipid nanoparticles (LNPs) for mRNA delivery in vivo. LNPs containing lipid M and encapsulating mRNA encoding influenza virus genes increase anti-influenza virus IgG titers in cynomolgus monkeys without inducing local edema, erythema, or systemic levels of IL-6.
More description
|
|
| DC67212 | Acuitas Lipid III-25 Featured |
Acuitas Lipid III-25 is an novel ionizable amine lipid used for mRNA delivery from Acuitas Therapeutics patent US 10,166,298 B2, with pKa 6.22, Liver Luc 1648 for 0.3mgkg(ng luc/g liver), Liver Luc 13880 for 1mgkg(ng luc/g liver) . It is an analgous of ALC-0315, showing higher activity than ALC-0315.
More description
|

|